Local
41 HIV, PrEP patients sue Gilead
They claim pharmaceutical company sought profits over patient health


(Gilead logo)
Pharmaceutical giant Gilead was hit with a lawsuit filed by 41 people from 12 states on April 11 who allege they suffered bone and/or kidney damage after taking Gilead’s tenofovir disoproxil fumarate, (TDF). The personal injury suit, filed in California Superior Court in Los Angeles, alleges the pharmaceutical company knew in 2001 that TDF was “highly toxic in the doses prescribed and risked permanent and possibly fatal damage to the kidneys and bones” —and that there was a safer alternative.
“More and more plaintiffs are coming forward to tell their stories of how they have been harmed by Gilead’s practice of putting profits over patient health,” said Liza Brereton with HIV Litigation Attorneys.
The suit is part of an ongoing effort to hold Gilead accountable for its alleged failure to rectify a known defect in the drug formulation of TDF, knowing that a safer alternate existed—tenofovir alafenamide (TAF). Additionally, AIDS Healthcare Foundation spokesperson Ged Kenslea told the Los Angeles Blade, the suit also includes Gilead’s failure to warn patients of TDF’s damaging side effects and its active misrepresentation of TDF’s efficacy and substantial risks. AHF is paying for the litigation and only seeks legal costs in return.
“Gilead had their safer alternative available and suppressed it in a malicious deliberate way for over 15 years just so they could maximize and extend the profits on TDF,” Kenslea said.
The suit also asserts that Gilead deliberately and maliciously kept information about TAF quiet to extend its patent, FDA exclusivity, and sales of its existing medications, including TDF. Gilead netted over $18 billion in profit in 2015.
The lawsuit alleges that “thousands and thousands of HIV/AIDS patients may have been unwittingly exposed to significant kidney and bone damage from TDF” during their antiretroviral drug regimens. Additionally, “many HIV-negative individuals seeking to prevent HIV acquisition may have suffered similar harm to their kidneys or bones from taking Truvada as part of their PrEP (pre-exposure prophylaxis) protocol.”
Gilead sells TDF under the brand name Viread. TDF is also a component of Gilead’s Truvada.
“Gilead knowingly continued selling a drug that had debilitating side effects while keeping a safer version in the wings until the patent on the first, more dangerous version had been exhausted. In other words, Gilead allowed serious injuries to occur so they could make a few more bucks before releasing a safer version,” prominent AIDS activist and “My Fabulous Disease” blogger Mark S. King told the Los Angeles Blade. “Meanwhile, Gilead continues to own our governmental and community-based HIV response lock, stock, and barrel by throwing endless cash at national organizations that should have our best interests at heart. Gilead’s behavior has been truly vile.”
King added: “I have been critical of AHF in the past for the quickness to sue, primarily other HIV organizations with which they differed. But this lawsuit is righteous and I hope they win, big time.”
Long Beach
Long Beach Pride canceled hours before start time, the community reacts
The City-funded and produced Pride Parade will continue as planned, but the community still wants answers

SoCal’s queer social media exploded into a frenzy when Long Beach Pride took to Facebook and Instagram to announce that the City of Long Beach had taken action to cancel this year’s Pride Festival, hours before Teen Pride, the kick-off event for the weekend, was to begin. Tonya Martin, President “Lez Prez,” called on the mayor and the City to move ahead with the Festival, using the current wave of national anti-LGBTQ sentiment as a reason to stand firm. The community wasn’t buying it and wanted more answers from Long Beach Pride.
In a statement made on social media, Martin shared, “Long Beach Pride is deeply disappointed by the City’s decision to cancel the Long Beach Pride Festival, a long-standing community institution built by volunteers, sustained by love, and rooted in the belief that every person deserves to live openly, safely, and with dignity.”
Martin appealed to queer activism as a reason for keeping the Festival going. “This decision comes at a moment when LGBTQ+ people are facing escalating attacks from the current federal administration and from political forces across the country. At a time when our community is being targeted and made vulnerable, Long Beach should be doing more to protect and uplift us, not taking away one of the most visible and meaningful expressions of inclusion our city has.”
Martin further called on the Mayor and the City to rethink the cancellation, “We call on the City of Long Beach to immediately engage in good faith with Long Beach Pride, community leaders, public safety partners, and elected officials to identify a path forward that preserves the festival and protects the community. We call on our Mayor Rex Richardson and the city council members to make the Pride Festival happen. We ask that our city leaders stand with the community at this critical moment and help ensure that Long Beach remains a beacon of equality, safety, and pride.”
It didn’t take long for the local news to show up.
The City was clearly being called out, the statement inciting the community to rally and demand the Festival take place. This year marks the 43rd Long Beach Pride. The Festival is a 100% volunteer organization, supported by allies, business owners, and the community at large.
Is the City the villain here? The City was quick to make an official statement, and the truth behind the cancellation became clear. “The Long Beach Pride Festival will not be able to take place this year as sufficient information to safely permit the event has not been made available by the event organizers.”
The statement further clarified that the decision was wholly due to the inability of the Festival to comply with City requirements, despite the fact that this event has taken place 42 times prior with the same needs in place.
“Over the past several months, the City of Long Beach’s Special Events team has worked closely with Long Beach Pride, the private organizers of the annual Pride Festival, to support their efforts to safely produce this year’s event, which was scheduled to take place on May 15, 16 and 17. While the City now manages and funds the Long Beach Pride Parade, the Pride Festival remains an independently organized, ticketed event that requires the submission of detailed operational, construction and public safety plans in order to be permitted to ensure safety of the attendees.
Despite continued collaboration and multiple deadline notices, the City did not receive the required documentation needed to complete safety reviews, inspect critical event infrastructure, such as the stage, electrical systems and tent, and emergency exiting plans to ensure compliance with public safety standards. With event programming scheduled to begin today, May 15 at 5 p.m. with Teen Pride and essential information still outstanding, there is no longer sufficient time to safely permit the festival this year.”
The statement also clarified that the Festival was alerted on Thursday that requirements had not been met, and that the City worked tirelessly with the volunteer organization up until the last minute, with the Festival still being unable to get everything in place. The City also made a promise to refund any businesses that had purchased special licenses or Health permits.
So the truth was out. The fact that the Festival intended to vilify the City and use the community’s spirit of activism to force the City’s hand in moving forward with a weekend that could be unsafe to attendees did not go over well. Social media comments on the Festival’s posts want more answers and they want the Festival to hold itself accountable. How has the Festival gone on for more than four decades prior without a cancellation, why this year? What happened? Yes, credit needs to be given for a volunteer organization to be able to produce the Festival year after year, but when an organization is 100% volunteer based, it is hard to hold people accountable.
But there are always at least two sides to a story. Local drag queen Twiggy D. Warhol took to social media to hold the Festival responsible.
An apparent committee member responded, saying all permits were sent:
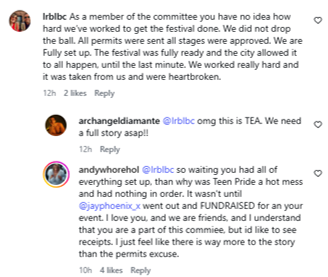
As all of this just came to light less than 24 hours ago, there will be more facts that will need to be shared from both the City and the Festival.
As much as local media picked up on the cancellation of the Festival, news channels and social media have also been promoting that the Pride Parade, funded and organized by the City of Long Beach, is still going on, along with five City-approved events.
Visit Long Beach posted a fun video, assuring the community that Pride is still going strong, despite the Festival cancellation. The bars will be open, featuring their own Pride programming and no doubt the streets will be flooded with the community proving that nothing can keep us down. ,
As many social media comments stated that Pride as a movement can never be canceled, and no one can ever take our spirit away. Where there is a will, there is a way. No doubt many more facts about the Festival cancellation will come to light. Perhaps this is the hiccup the Festival needs to reorganize and revitalize. And maybe this is the hiccup the Festival needs for the community to see that it needs more support from us to ensure this doesn’t happen again.
See you at the Pride parade!
Los Angeles
Los Angeles Metro officially opens D Line extension through Mid-City; Mayor Karen Bass claims it’s ‘built for the future’
U.S. Rep. Laura Friedman called out the nation’s capital for their “attack” on California and public transportation

Getting around our car-infested city just got a little bit easier.
On Friday, May 8, hundreds of Metro board members and Los Angeles County officials gathered at the Petersen Automotive Museum to celebrate the D Line’s new service to three stations. It’s the latest effort – albeit a long overdue one – to expand L.A. public transportation through the heart of the city. LA Blade was on the scene.
With stations connecting Western Avenue to La Cienega Boulevard, the new line now makes travel from Beverly Hills to Downtown Los Angeles possible in 21 minutes, according to Metro officials. To celebrate the project’s commitment to connecting people to culture, the ribbon-cutting ceremony was followed by a celebratory outdoor event with local food vendors.
CEO Stephanie Wiggins addressed why the L.A. Metro “might just be having a moment” right now: “When gas prices are off the charts, traffic feels endless and parking costs are more than your lunch – $1.75 a ride that gets you pretty much anywhere in the region starts to look pretty smart.”
Getting to West Hollywood by transit – a vital spot for the city’s queer culture and nightlife – is another goal for the county. But there’s still a ways to go: construction on an official station isn’t expected to start until 2041.
While the D Line expansion was made possible by a grant from the federal government, L.A. Mayor Karen Bass said at the press conference that “we don’t wait for the federal government. We know we can’t depend on others. Obviously, we need the federal government and the state government, but Angelenos say we are going to tax ourselves to do what is needed.”
This expansion comes as L.A. invests billions of dollars in public transportation ahead of the 2026 FIFA World Cup, 2027 Super Bowl, and 2028 Olympics – all events placing an extra spotlight on one of the busiest cities in the country. Just last June, a metro stop opened at LAX, connecting residents and visitors to L.A’s main airport.
As L.A. tries to catch up with the public transportation infrastructure of major hubs like New York City, one speaker also took the opportunity to address tensions with the nation’s capital:
“I will tell you, being in D.C., there is such an attack against California coming from Washington right now, and against transit across the nation,” U.S. Rep. Laura Friedman said. “And we are here to show Washington that while you know who is tweeting at 3 a.m., we have Angelenos underground building things for this country. We are showing that transit is essential, and we are not going to let Washington defund our children’s future.”
Ongoing construction for the L.A. Metro continues as the city hopes to make big on its “28 Projects by 2028” promise. But for now, residents can enjoy free rides all weekend long, along with 60 days of activations and events at the three new stations.
“I am looking forward to being at the ribbon cutting in a few more years as we move forward and take this line all the way to Westwood,” Bass said. “We celebrate something that Angelenos deserve – a city that’s easier to move through, easier to connect in, and that’s built for the future.”
Los Angeles
LA LGBT Center’s first legacy cycling event raises over $800K
From April 24-26, 300 cyclists rode from Los Angeles to San Diego, raising funds and awareness for the Center’s LGBTQ+ serving programs.

On Friday, April 24, 300 people gathered just before dawn, rolling their bicycles to a stop in Elysian Park. Against crisp morning air and dark, they donned vibrant orange and pink athletic wear, protective helmets, and sunglasses; while the rest of the city remained sleepy, the large group, which grew larger by the minute, hummed with excitement as they prepared to take off together on a three-day adventure towards the San Diego LGBT Community Center.
This was the start of the highly anticipated Center Ride Out, the first-ever AIDS/LifeCycle legacy event created by the Los Angeles LGBT Center. For over 30 years, AIDS/LifeCycle brought masses for a seven-day ride from San Francisco to L.A. and raised over $300 million for life-saving HIV and AIDS resources and services.
Center Ride Out was built in the lasting imprint and shadow of this event, and strived for a more accessible and joyous approach. “Center Ride Out carries forward the legacy of AIDS/LifeCycle, rooted in a time when our community came together to care for one another,” said LA LGBT Center CEO Joe Hollendoner, in a press release, who describes Center Ride Out as the beginning of a new legacy for LGBTQ+ cycling activism.
Described as a “queer summer camp,” the pared-down three-day journey began with a 110-mile trek towards Temecula’s Lake Skinner, where, after a night’s rest, cyclists could spend a day gathering with community over arts and crafts, massages, a dance party, games, and other activities reminiscent of summers spent simmering by the water.
On the third day, cyclists rode 87 miles to the San Diego LGBT Community Center, one of the event’s benefiting partners, rounding out a nearly 300-mile journey across Southern California. In total, cyclists raised $830,511 to support the LA LGBT Center’s vital LGBTQ+ healthcare, housing, educational, and advocacy programs and social services — a crucial accomplishment after the organization suffered a $9 million loss in federal funding in the last fiscal year.

Sunday marked a victorious end to this first iteration of Center Ride Out, and cyclists raced towards each other upon reaching their final destination: sweaty, tired bodies embracing and entangling in pride and accomplishment. The monumental AIDS/LifeCycle has come to an end, but the joy that reverberated from this evening signaled the start of something just as great.
Registration has already begun for the next Center Ride Out, which returns April 23-25, 2027. Learn more at the Center’s website.
Kristie Song is a California Local News Fellow placed with the Los Angeles Blade. The California Local News Fellowship is a state-funded initiative to support and strengthen local news reporting. Learn more about it at fellowships.journalism.berkeley.edu/cafellows.
Los Angeles
LGBTQ+ mayoral candidate wants to revitalize a ‘limitless’ L.A. of the past
There’s no clear frontrunner in L.A.’s mayoral primary. Will queer candidate, Bryant Acosta, shake things up?

We are just a little over a month until the Los Angeles primary mayoral election takes place on June 2.
Incumbent mayor Karen Bass, District 4 councilmember Nithya Raman, and conservative reality star Spencer Pratt currently lead the race, but a poll released early this month by the UCLA Luskin School of Public Affairs indicates unsteadiness and volatility ahead. 40% of voters remain undecided — and without a majority vote, two leading candidates will have to face off in a runoff election on Nov. 3.
Some are hoping for an underdog to swoop in, and the Blade spoke with one of these contenders: LGBTQ+ artist and creative director, Bryant Acosta.
Born in West Covina, Acosta attended The Art Institute of California, Los Angeles in the early 2000s, where he experienced a bursting energy that defines the county’s reputation as a major hub and global beacon for success. “Everything felt limitless. There was so much opportunity,” Acosta told the Blade. “The city felt alive. You could feel the heart and soul of what L.A. was, and I just don’t see that anymore.”
Acosta hopes to revitalize the county with fresh perspectives, a creative tech-forward approach, and a reboot of how City Hall operates when it comes to transparency and efficiency. His career has spun through several evolutions: he’s been in charge of multi-million dollar budgets and large teams as a creative director, both in the corporate world and on his own terms.
The Blade sat down with Acosta to discuss his pledge for the county’s queer residents and other minority community members, his vision for an app that would streamline accessible city services, and how he sees his identity as a ‘superpower.’
What are your ideas about how you would concretely support queer communities? I was at City Hall a couple months ago when the TransLatin@ Coalition was there to ask for $4 million in direct funding from the county. There’s a big wave and throughline of struggle when it comes to queer communities having to advocate for themselves. How would you support them as Mayor?
Being queer myself…I just feel like you don’t have to shrink yourself to survive in the city, at least not on my watch. This campaign is for the people who have felt unseen, who have had to build their own community and opportunities. But it isn’t just about visibility, it’s about affordability, safety, and opportunity. Because what good is being seen if you can’t afford to live here or feel protected?
Education [is] a big [priority] because right now there’s so much misinformation — specifically around trans issues. I want to be able to bring people along on the journey of: Hey, this is who they are, this is what they’re asking for [and] make it so that people see them as humans. Being able to bridge that gap between the misinformation on social media and bridge it to actual science-based information so that people can really understand what it is to be trans and that they’re part of our community.
When I spoke with trans leaders and advocates, many explained that City Hall does not lack funds — it’s simply not prioritizing their organizations into the county’s budget.
I know Kenneth Mejia, our [City] Controller, has been working on having more visibility into the budget — but if we were able to have an application like my LA Now app idea where people can track every dollar [and] every penny spent, it’s not going to be a fight. This minority group of people is asking for this much money. It’s well within the budget. Why wouldn’t we do this? It’s an essential service for them. Just as the trash or the water are essential services for your neighborhoods, these are things that they’re asking for to continue to operate and be a part of our community. That’s why I’m making that my main touch point, because without accountability, transparency and trust in City Hall, we don’t have anything else.
Can you tell me more about your LA Now app idea and how it adds to your mission of transparency for LA county residents?
I worked in tech for a while, so I learned how to use technology to better…communications, advertising, and all of the things. So, I [thought]: I should be able to use that and be a really future-forward mayor.
I developed this app, where basically, you could have civics in the palm of your hand. You’d open the app, [and] you’d have a dashboard. Organizations will be loaded into it so that when you have problems with housing, the streets and anything in your community, it’ll geo-target your area so they’ll have all the services listed that you can contact.
You’d also be able to pay parking tickets. If you get towed by the city, it’ll give you a notification. So, there’s no more of those predatory towing fees. [You’d also be] able to get push notifications for jury duty, so it has a lot of those civics built in.
And with what Kenneth Mejia is doing right now — he’s giving us a full data dashboard of where the money’s going. I want to simplify that and make it more accessible so that, [for example], my mom and my cousins can read it. [I want to] really put it in the palm of your hand so we see every dollar, every penny spent in real time.
I created a section where, essentially, you can rate your leader. So basically it’d be like Yelp, but for leadership. So, when you see things like Nithya [Raman] spending a million dollars on bathrooms, that would trigger a warning to the Controller, and then we’d be able to see: Hey, what’s going on here? If leaders want to have five stars, they need to respond in real time. It would just keep that extra layer of accountability — a digital accountability on leadership.
Last but not least, I want to develop an anti-Amazon feature [on the app]. Essentially, we would have an E-commerce marketplace [that] only local businesses would be able to get on, so that they could maximize their profits.
Would the app include surveillance protections? With fears over ICE and online safety, would there be a way for people to feel secure while using the app?
How I’m thinking of this as an operating system is that we would use ID.me, which we use at the DMV, to be able to log in. So it already has all your information, and that way you’re not having to input everything a million times. But for people who are undocumented, you’d have a back portal where you’d have access to essential services. I [also] thought about people who don’t have cell phones. We could also have kiosks in public libraries, grocery stores, any place that has public access, so that everybody would have access to these data points and things that they may [need].
You’re forward about being at the intersection of marginalized identities, as a queer and Latino person. How does this affect how you think about the mayoral race and how you’re building connections with LA residents?
Being queer and Latino [doesn’t] hold me back. It’s actually a superpower for me. In some ways, it forced me to figure things out — not just to find a seat at the table, but to build my own. Understanding people [in] these kinds of ways is invaluable. Also, being first-generation American and openly gay, you see this country as both an opportunity and an exclusion at the same time. No matter how smart, accomplished, or creative you are, there’s still a ceiling you hit, especially when the system was never built with you in mind.
That perspective really changes how [I] lead because you don’t just want access to the system — you want to fix it so that it actually works for everyone.
To learn more about Bryant Acosta and his mayoral campaign, you can visit his social media page and website.
Kristie Song is a California Local News Fellow placed with the Los Angeles Blade. The California Local News Fellowship is a state-funded initiative to support and strengthen local news reporting. Learn more about it at fellowships.journalism.berkeley.edu/cafellows.
West Hollywood
Lesbian cinema, from the archives and beyond, lead this short film festival
This Saturday, the June L. Mazer Lesbian Archives celebrates Lesbian Visibility Week with archival and contemporary sapphic shorts.

At the June L. Mazer Lesbian Archives in West Hollywood, Alisha Graefe and Kymn Goldstein are rummaging through a treasure trove of unknown lesbian films. Large, neon orange bins sit at their feet, filled to the brim with VHS tapes donated by queer filmmaker Rosser Goodman, who in the late 90s and early 2000s programmed the underground lesbian screening series “Film Fatale.”
Her monthly event provided a safe haven and experimental ground for lesbian film in L.A., shining light on new voices, stories, and perspectives that explored the nuances of living life as a queer woman.
This Saturday, April 25, Graefe and Goldstein are paying homage to Goodman’s legacy with a cinema-centered event of their own: a short film festival that brings together an eclectic, curated mix of archival works and newly submitted pieces from emerging lesbian filmmakers around the world. These works, accompanied by panel discussions, will screen all day in three different blocks at the Pacific Design Center’s Silver Screen Theater.
Like “Film Fatale,” this festival is not just a showcase of resonant, timely art: it’s an opportunity for queer people to intentionally gather, reflect on their history, joy, and resilience, and to soak in spaces that celebrate them.
“The archives are full of history, but every single day we make history,” Goldstein, who is the archive’s executive director, told the Blade. “This festival is an act of creating history. [We’re] bringing people together to celebrate Lesbian Visibility Week and to watch these films. [And] you know what, every week is Lesbian Visibility Week at the archive.”

When they were first planning out the film festival together, Goldstein and Graefe — who serves as the Mazer’s full-time archivist — sifted through Goodman’s collection, letting the sounds of static, VCR clicking, and tape-scrubbing fill the room. One of the films they watched, and which will screen at Saturday’s festival, is Goodman’s own 14-minute short, “Life’s a Butch!”: a silent comedy about a woman’s antics to impress her new femme crush with clumsy, masc charm.
It was made over 25 years ago, but its tenderness and whimsy are still palpable today. This, and the other films in the festival, speak to both singular and universal emotions and experiences lesbian, queer, and sapphic people have experienced and will continue to experience across time and space.
This interconnectedness in films, communities, and shared memories between past and present excites Goldstein. “The stories of coming out, of crushes, of losing love…[They] are the same topics and subjects that come up today, especially the political things we’re going through. There’s a continuity across all of it.”
And where there’s continuity, there are clues and maps that pinpoint paths of resistance, dialogue, and survival. When people see their lives, or the lives of others in their communities, reflected on screen and in narratives they can interact with, they are able to draw upon history to craft their own ways forward.
The film festival also offers local community members an entryway into the archive, a longstanding community space that houses journals, photographs, books, films, letters — all tangible materials that people are encouraged to touch and engage with, whether it’s to further their research, spark ideas, or simply be more intimately in conversation with the past.
“There are tons and tons of stories,” Goldstein said, who is focused on maintaining and growing the archive’s collection of ephemera and other personal materials from the community. “It’s living, breathing history in a variety of forms, all hidden in different-sized boxes that you just have to open to see what’s inside.”
To support and learn more about the archive’s upcoming film festival, collections, and other events, visit their website.
Kristie Song is a California Local News Fellow placed with the Los Angeles Blade. The California Local News Fellowship is a state-funded initiative to support and strengthen local news reporting. Learn more about it at fellowships.journalism.berkeley.edu/cafellows.
Arts & Entertainment
LA Blade’s Best of LA 2026 winners announced!
The top names from SoCal’s LGBTQ community, as voted on by LA Blade Readers, are honored for the 9th annual Best of LA.

This week marks the Los Angeles Blade’s 9th Annual Best of LA Award Show, honoring leading members from the community as voted on by readers of the Blade. Presented in part by MISTR and Visit West Hollywood, the community gathered on Thursday, March 26th, at The Abbey, to celebrate the winners:
Best Drag Performer
Cake Moss
Kyra Jete, Runner-up
Best Drag Show
Rocc-ettes at Mattie’s
Hamburger Mary’s Weho. Runner-up
Local Influencer of the Year
Rose Montoya
Lucas Dell, Runner-up
Best LGBTQ Bar
Gym Bar
The Abbey, Runner-up
Best Happy Hour
Motherlode
Hi-Tops, Runner-up
Go-Go of the Year
Steven Dehler
Prince Joshua, Runner-up
Best Restaurant
Bottega Louie
WeHo Bistro, Runner-up
Best Radio or TV Station
REVRY
KTLA, Runner-up
Best Cannabis Retailer/Lounge
Green Qween
Artist Tree Lounge, Runner-up
Best LGBTQ Owned Business
MISTR
Green Qween, Runner-up
Best LGBTQ Social Group
Gay Men’s Chorus of Los Angeles
Dark Circle Film Society, Runner-up
Best House of Worship
Hollywood United Methodist
Congregation Kol Ami, Runner-up
Activist of the Year
Maebe A. Girl
Rose Montoya, Runner-up
Public Official of the Year
John Erickson
Lindsey Horvath, Runner-up
Best Local Pro Sports Team, co-presented by Pride House LA/West Hollywood
LA Dodgers
LA Lakers, Runner-up
Local Ally of the Year
Kevin De Nicolo
Jessica Steinman, Runner-up
Best Doctor/Medical Provider
AIDS Healthcare Foundation
LA LGBT Center, Runner-up
Most LGBTQ-Friendly Workplace
Los Angeles LGBT Center
City of West Hollywood, Runner-up
Non-Profit of the Year
Los Angeles LGBT Center
Trans Lifeline, Runner-up
Best Local Actor
Annie Reznik
Matthew Scott Montgomery, Runner-up
Best Local Theatre
Celebration Theatre
Pasadena Playhouse, Runner-up
Local Musical Artist of the Year
Prince Joshua
Ross Alan, Runner-up
Best LGBTQ Event
Outloud Music Festival at Weho Pride
GLAAD Awards, Runner-up
Best Regional Pride
WeHo Pride
Long Beach Pride, Runner-up
Best Promoter of the Year
Beau Byron
Paul Nicholls, Runner-up
LGBTQ Professional of the Year
Erik Braverman
Michael Ferrera, Runner-up
Best Bartender
Danny Hernandez
Michael Susi, Runner-up
Best DJ
Boy Apocalypse
DJ Les Ortiz, Runner-up
Best Local LGBTQ Podcast
BabyGay
On The Rocks, Runner-up
Best Salon/Spa
Shorty’s Barber Shop
The Massage Company WEHO, Runner-up
Best Music Venue
The Hollywood Bowl
The Troubadour, Runner-up
Best Fitness/Workout Spot
LA Fitness, Hollywood
Barry’s WEHO, Runner-up
Best Hotel
Hotel Ziggy
SoHo House, Runner-up
This year’s Local Hero Award goes to Genevieve Morrill in honor of her role as an ally, serving as president and CEO of the West Hollywood Chamber of Commerce.
Gus Kenworthy received the first-ever Los Angeles Blade Athlete Legacy Award, co-presented by Pride House LA/West Hollywood.
Los Angeles Blade publisher Alexander Rodriguez shared, “In light of the year we’ve had to contend with as the queer community, it is imperative that we take moments to share the accomplishments and resilience of our local family of activists, leaders, and allies. Honoring our nominees and winners this year is proof of the power of queer joy. Until next year!”
Los Angeles
The Connie Norman Transgender Empowerment Center opens full-service health clinic
Saturday marked the opening of the center’s new health clinic and another step forward in its “righteous rebellion” for trans equality.

It’s warm on Saturday morning, and there’s joy in the air at the Connie Norman Transgender Empowerment Center (CONOTEC). By 10 a.m., community members have packed out the center’s first floor, waiting eagerly for the grand opening of its new healthcare clinic. It’s a historic moment that flows from the core of CONOTEC’s purpose: advocating for, creating pathways and uplifting the safety, resilience and wellbeing of trans and queer people.
Established by the AIDS Healthcare Foundation (AHF) in 2021, CONOTEC honors the legacy of its revered namesake, the “AIDS diva” Connie Norman: a trans activist who was at the frontlines of fighting against government inaction and negligence during the AIDS crisis of the 1980s. For five years, the center has been the home base for various local trans and queer organizations, providing a sanctuary for queer elders and advocates to expand and continue their advocacy.
Now, the center’s new health clinic takes this mission further. A collaborative effort between trans medical professionals, activists, and AIDS Healthcare Foundation leaders, the clinic offers an expansive breadth of services that covers primary care, HIV medicine and treatment, sexual health, cancer screenings, and vaccinations. It is open to everyone, regardless of gender and sexual identity, as well as one’s ability to pay.

Advocates name the last few years as a major setback to trans health care, safety, and rights. As the federal administration targets this community by shuttering gender-affirming health clinics, writing laws that approve surveillance of and violence against trans people, the opening of this clinic is both an act of resistance and care. “We get to open doors instead of watching them close,” said Queen Chela DeMuir, who is vice president of AHF’s trans affinity group FLUX and the chief operating officer at CONOTEC.
Addressing the room with tenderness, DeMuir recounted the beginning of her transition. Her first hormone shot was administered by a queer elder she trusted, the medicine bought from a shop at a local swap meet. She remembered the confusion, the concern, the absence of professional care. But, like many other trans folks, she was willing to take these risks to actualize and make physical the person she always was.
The clinic removes the “guessing, fear, and barriers” DeMuir experienced, making sure trans people can receive the care they need in safe, affirming, and accessible spaces. “In this clinic…we care for everyone. We are building access with people who look like us, who understand us, and who carry a shared, lived experience…This is not just a ribbon-cutting. It’s a statement about who we are and what we deserve,” said DeMuir.

What trans and nonbinary community members deserve is easy access to healthcare and, thus, easy access to “justice, dignity and the right to live fully,” stated AHF chief medical officer Dr. Carl Millner. He reiterated that trans people, specifically trans women, are disproportionately impacted by HIV yet have fewer pathways to access care and support.
The clinic rewrites standard medical practice by centering trans lives in both their patient outreach and in their staff. Razan Alawadhi, an AHF nurse practitioner and trans woman, will bring nearly two decades of healthcare experience to help guide the clinic’s operations. “It is a dream of mine to establish this clinic,” said Alawadhi, who also hopes to advance studies and treatment in neglected research areas like sexual health for trans people.
On the other side, trans people who seek care are repeatedly met with varying levels of hostility and misunderstanding in health care spaces. “I remember going to an emergency room for a back injury — something unrelated to my transness — and being looked at like a freak,” said FLUX ambassador Laith Ashley, who described the humiliation with crystal clear detail. Those experiences don’t fade away; they leave lasting marks and create cycles of medical avoidance in trans and nonbinary communities.
“This [clinic] is here to help change experiences like that,” Ashley continued. “Here…providers recognize that affirming someone’s identity is not controversial. It’s compassionate medicine…So when we open a clinic like this, we are not just expanding health care services. We are expanding hope [and] sending the message that transgender people in this community are not alone.”
This message is not new. The fight for freedom was forged by trans ancestors, and is carried onward by present and future generations. It’s a “full-circle moment,” DeMuir explained to the Blade. “I knew that I was putting myself at risk when I was younger, so part of the work that I do is really making sure we make things better for those that come [after] us, and give them a sense of pride that they are seen.”
The fire beneath this fight for visibility blazes with the warmth and strength of its leaders and community. It’s just beginning. “I want to welcome you to the righteous rebellion,” said Queen Victoria Ortega, president of FLUX and the chief visionary officer of CONOTEC. “[This is] a place where doors will be open for folks…No one’s going to deport us. No one’s going to erase us off the earth. This is a place for [us] to have dignity and have access to health care for all.”
More information about the Connie Norman Transgender Empowerment Center can be found on their website.
Kristie Song is a California Local News Fellow placed with the Los Angeles Blade. The California Local News Fellowship is a state-funded initiative to support and strengthen local news reporting. Learn more about it at fellowships.journalism.berkeley.edu/cafellows.
California
Equality California has sponsored 12 bills to advance LGBTQ+ rights in the state
On Feb. 27, the LGBTQ+ civil rights organization announced its 2026 legislative priorities, which cover trans healthcare and queer education.

Nationally, queer and trans people are facing a crisis. Last July, the Trump administration defunded the 988 suicide prevention lifeline for LGBTQ+ youth. Anti-trans hate crimes are on the rise, specifically those targeting Black and brown trans women. Just last week, trans people living in Kansas who had changed their gender designations in the past received abrupt letters stating that their driver’s licenses were no longer valid.
Federal and statewide legislation is targeting LGBTQ+ rights, creating a social landscape that is reverting to sanctioned violence against these communities. In response, LGBTQ+ civil rights organization Equality California has sponsored 12 new bills that are fighting to strengthen queer people’s rights, safety, and sense of stability.
This means “being able to access health care, live free from discrimination, gather safely, and trust that our personal information will not be weaponized against us,” said Equality California’s executive director, Tony Hoang, in a press release. Hoang also states that these bills offer Governor Newsom an “opportunity to define real leadership” and set himself and the state apart from the proliferation of anti-LGBTQ+ rhetoric amongst the most politically elite.
What are the bills and what would they do?
The 12 priority bills Equality California has sponsored span various areas, from healthcare to the criminal justice system and from schools to emergency hotlines.
AB 1876 (Dawn Addis): Protecting transgender patients from discrimination
Introduced on Feb. 12, AB 1876 would prohibit health insurers and plans from discriminating against people based on sexual orientation, gender identity, or sex characteristics. This would help advance the rights of trans, gender diverse/expansive, and intersex (TGI) people across California, allowing them greater accessibility when seeking the healthcare they need.
AB 1930 (Rick Zbur): Protecting transgender patient privacy from out-of-state investigations
Introduced on Feb. 13, this bill was written by Assemblymember and former Equality California executive director Rick Zbur. It focuses on protecting transgender patients who have received gender-affirming care, abortions, or any other form of trans health care in California. AB 1930 will place greater protections on confidential, private medical information, barring out-of-state agencies from readily obtaining these records and using them against trans people.
SB 1114 (Christopher Cabaldon): Protecting LGBTQ+ data privacy
California-based agencies continue to champion research on LGBTQ+ communities, collecting data related to gender and sexual identity. However, there are concerns that federal officials will try to surveil, shut down, or extract this data. SB 1114 would counter this by limiting when state agencies can share this sensitive data and barring the disclosure of such data outside of California.
AB 1540 (Mark González): Restoring the youth crisis support hotline
Since last year, after the administration ended the 988 Suicide and Crisis Lifeline’s specialized services for queer youth, L.A. officials have been advocating for its revitalization. AB 1540 was introduced this January by prominent LGBTQ+ Assemblymember Mark González, and the bill calls for the restoration of the “Press 3” option for queer youth struggling with their mental health. Many have lauded the service’s necessity, urging officials to recognize and take action to address the broader mental health crisis of young LGBTQ+ people across the country.
SB 934 (Scott Weiner): Advocating for conversion therapy survivors
SB 934 aims to extend the statute of limitations so conversion therapy survivors can pursue legal claims against licensed mental health professionals who have subjected them to conversion therapy practices.
In other states, victories against conversion therapy have taken a step backwards. In 2023, conversion therapy was legally banned, to prevent the “horrific practice” from continuing to harm LGBTQ+ youth. This past December, the ban was blocked, on the grounds that it violated First amendment rights for therapists and counselors — even if they engaged in the practice.
AB 1775 (Chris Ward) — Supporting transgender veterans
In January 2025, the Trump administration issued an executive order that barred trans people from serving in the military, stating that the forces had been “afflicted with radical gender ideology.” As a result, tens of thousands of active-duty service members, veterans, and their family members were abruptly cut off from a steady income, putting them at risk of homelessness and financial instability. AB 1775 will make sure trans veterans impacted by the executive order can access housing assistance, employment support, and be able to correct their military orders.
AB 1836 (Jesse Gabriel): Protecting safety of LGBTQ+ community events
This bill intends to expand the state’s Nonprofit Security Grant Program so that organizations can access funding for security efforts when hosting LGBTQ+ community events. Currently, the grant program provides funding for LGBTQ+ community centers and nonprofit facilities, but does not cover off-site events. For queer people celebrating their joy in public, which can be its own act of resistance, the possibility of retaliation leaves in many a remnant of fear and apprehension. This bill combats these fears by creating funding opportunities to ensure proper protections can be accessed at such events.
SB 1023 (John Laird): Creating greater access to injectable PrEP
Assemblymember Laird’s bill proposes improved insurance coverage and reimbursement practices so that healthcare providers can offer long-acting injectable PrEP without being slowed down by financial or administrative barriers.
Community clinics often face the brunt of ineffective insurance reimbursement practices, which blockade and clog their ability to provide HIV preventative medicine. SB 1023 aims to make these processes smoother, ensuring that clinics and providers can provide PrEP in a timely, equitable manner.
AB 908 (Jose Solache): Ensuring LGBTQ+ curriculum is meaningfully adapted
This bill would require California’s Department of Education to make sure school districts are complying with the state’s FAIR Education Act, which calls for curricula to cover the histories and contributions of various marginalized communities, including Black, brown, indigenous, and LGBTQ+ people.
The FAIR Education Act was signed into law in 2011, but AB 908 calls attention to the fact that only 37% of school districts actually implement meaningful LGBTQ+ education in their curricula. The bill reaffirms what has already been legally mandated for 15 years: that school systems concretely cover LGBTQ+ history and contemporary advancement for their students.
SB 1328 (Sabrina Cervantes): Creating more LGBTQ+ outreach for higher-ed students
This bill would require higher education institutions to designate specific confidential employees at satellite campuses, outreach centers, and other external branch facilities to support the needs of LGBTQ+ students and staff.
LGBTQ+ “points of contact” can be found at various primary campuses, and this bill calls for the same measures to be taken at outreach centers related to higher education institutions. SB 1328 argues that more should be done for queer students, faculty, and staff who are present at an institution’s various locations — not just its main campus. It highlights a need for intention and care for queer people’s needs within the education system: cursory attempts at inclusion are not enough.
AB 2014 (Sade Elhawary) — Preventing gender bias in criminal trials
The California Committee on Revision of the Penal Code released its 2025 annual report in December and identified that an important point of reform was the way gender-based evidence is treated in court. AB 2014 would require courts to apply heightened scrutiny — a more demanding form of review that requires substantial evidence — before hearing arguments based on harmful gender-based stereotypes.
SB 1149 (Maria Durazo) — Expand Bereavement Leave for Chosen Family
This bill expands the definition of work-protected bereavement leave to include chosen and extended family members. Currently, California law restricts bereavement leave to immediate family, and does not take into account that queer people are often piecing their own families together after abandonment, estrangement, or fear for their safety and health.
The pain of losing a chosen family member can be just as, if not more, searing and altering than the loss of a blood relative. SB 1149 would protect workers experiencing this kind of grief.
Kristie Song is a California Local News Fellow placed with the Los Angeles Blade. The California Local News Fellowship is a state-funded initiative to support and strengthen local news reporting. Learn more about it at fellowships.journalism.berkeley.edu/cafellows.

The 2026 Los Angeles Blade Best of LGBTQ LA Awards are here! You submitted your nominations—now it’s time to vote for the finalists. Voting is open through March 6, 2026.
Among some of your favorite categories are Best Drag Performer, Local Influencer of the Year, Best Happy Hour, Go-Go of the Year, Activist of the Year, Public Official of the Year, Best Non-Profit, Best Bartender, Best DJ, Best Local Podcast, and so many more!
Winners will be revealed at the Best of LGBTQ LA celebration on Thursday, March 26 at The Abbey. Stay tuned for more party details coming soon!
Vote using the form below or by clicking HERE.
Here are this year’s nominees!
Best Drag Performer
- Cake Moss
- Charles Galin King
- Kyra Jete
- Laylah Amor
- Misty Violet
Best Drag Show
- Bring It To Brunch at Mattie’s
- Brunch Service at The Abbey
- Hamburger Marys West Hollywood
- Las Reinas at Mickys
- Rocc-ettes at Mattie’s
Local Influencer of the Year
- Charles Hernandez (CnoteLA)
- Curly Velasquez
- Justin Martindale
- Lucas Dell
- Rose Montoya
- Victoria Pousada Kreindler
Best LGBTQ Bar
- Gym Bar
- Kiso Los Angeles
- Mattie’s Weho
- Or Bar
- The Abbey
Best Happy Hour
- 33 Taps
- Fiesta Cantina
- Hi-Tops
- Mickys
- Motherlode
- The Abbey
Go-Go of the Year
- Daniel Mooney
- Gabriel Gonzalez
- Jay Nova
- Prince Joshua
- Steven Dehler
- Victoria Shaw
Best Restaurant
- Bottega Louie
- Hamburger Mary’s
- La Boheme
- Pura Vita
- WeHo Bistro
Best Radio or TV Station
- CHANNEL Q
- KTLA
- LatiNation
- Out TV
- REVRY
Best Cannabis Retailer/Lounge
- Artist Tree Lounge
- Elevate
- Green Qween
- Med Men
- The Woods WeHo
Best LGBTQ Owned Business
- Fan Girl Cafe
- Green Qween
- JJLA
- MISTR
- Wildfang
Best LGBTQ Social Group
- Dark Circle Film Society
- Gay Men’s Chorus of Los Angeles
- NLGJA Los Angeles
- Outloud Sports
- Unique Woman’s Coalition
- WeHo Dodgeball
Best House of Worship
- Congregation Kol Ami
- Founders Metropolitan Community Church Los Angeles
- Hollywood Boulevard Episcopal
- Hollywood United Methodist
- InVision Church Los Angeles
Activist of the Year
- Cory Allen
- Joshua Marin-Mora
- Liliana Perez
- Maebe A. Girl
- Rose Montoya
Public Official of the Year
- CA State Treasurer Fiona Ma
- Chelsea Byers
- John Erickson
- Lindsey Horvath
- Maebe A. Girl
Best Local Pro Sports Team
- Angel City FC
- LA Chargers
- LA Dodgers
- LA Lakers
- LA Rams
- LA Sparks
- Los Angeles FC
Local Ally of the Year
- Abbe Land
- Jessica Steinman
- Kevin De Nicolo
- Lindsey Horvath
- Senator Lena Gonzalez
Best Doctor/Medical Provider
- AIDS Healthcare Foundation
- Better U
- Dr. Eric Chaghouri
- LA LGBT Center
- St. John’s Wellness
- UCLA CARE Center
Most LGBTQ-Friendly Workplace
- AIDS Healthcare Foundation
- City of West Hollywood
- JJLA
- Los Angeles LGBT Center
- Revry
Non-Profit of the Year
- AJ Socal
- Equality California
- Los Angeles LGBT Center
- OutAthletes
- Project Angel Food
- Trans Lifeline
Best Local Actor
- Annie Reznik
- Jason Caceres
- Michael Scott Montgomery
- Nhut Le
- Shaan Dasani
- Trevor Dow
Best Local Theatre
- Celebration Theatre
- Center Theatre Group
- Geffen Playhouse
- International City Theatre
- LA Opera
- Pasadena Playhouse
Local Musical Artist of the Year
- Prince Joshua
- Robert Rene
- Ross Alan
- San Cha
- Tom Goss
Best LGBTQ Event
- Dinah Shore
- GLAAD Awards
- LA Opera Pride Night
- MISTR’s National PrEP Day
- Outloud Music Festival at Weho Pride
- Pride Night by Hyperion LA
Best Regional Pride
- DTLA Proud
- Hermosa Beach
- Long Beach Pride
- Palm Springs Pride
- WeHo Pride
Best Promoter of the Year
- Andres Rigal
- Ash Rodriguez
- Beau Byron
- Joshua Flores
- Paul Nicholls
LGBTQ Professional of the Year
- Cory Allen
- Erik Braverman
- Kathleen Rawson
- Liliana Perez
- Michael Ferrera
- Tristan Schukraft
Best Bartender
- Alex Satoshi DiDio
- Danny Hernandez
- Manny De Cielo
- Matt Stratman
- Michael Susi
- Michael Vega
Best DJ
- Boy Apocalypse
- DJ Les Ortiz
- DJ SRO
- Lord Izac
- Simon Harrison
Best Local LGBTQ Podcast
- BabyGay
- No Matter What Club
- No Matter What Recovery
- On The Rocks
- Sloppy Seconds Podcast
- Very Delta
Best Salon/Spa
- Bautis LA
- Folklore Salon & Barber
- Project Q
- Shorty’s Barber Shop
- The Massage Company WEHO
Best Music Venue
- The Disney Concert Hall
- The Hollywood Bowl
- The Roxy Theatre
- The Troubadour
- The Wiltern
Best Fitness/Workout Spot
- Barry’s WEHO
- Equinox on Sunset
- Gold’s Gym
- John Reed Fitness
- LA Fitness, Hollywood
Best Hotel
- Andaz
- Edition Hotel
- Hotel Ziggy
- Kimpton La Peer Hotel
- SoHo House
California
Experts discuss pathways forward as anti-trans violence continues to rise
On Thursday, Feb. 19, the Williams Institute invited a panel of local experts to discuss the rise in anti-trans hate crimes, and ways communities can seek refuge and support.

During a recent webinar hosted by the Williams Institute, a local LGBTQ+ policy think tank, several policy experts, law scholars, and advocates gathered online to discuss violence against transgender people in California as well as potential solutions to navigate the year ahead.
Here are important updates gathered from the session. These expand on an earlier Blade article about the increase in reported hate crimes and incidents against trans people since 2013.
What we’re familiar with: trans people face higher rates of victimization and violence
llan H. Meyer, the Williams Distinguished Senior Scholar of Public Policy at the Williams Institute, utilized data collected by the 2022-2023 National Crime Victimization Survey (NCVS) and the 2022 U.S. Transgender Survey (USTS) to re-solidify the lived experiences of trans Angelenos and Californians.
The reality is: transgender, gender expansive, and intersex (TGI) communities face much higher rates of violence compared to cis people who are not queer-identifying.
Out of the 9,146 Californian respondents who participated in the national USTS, 19% of those surveyed reported that they received threats of violence. 38% reported facing verbal harassment, and 42% experienced online harassment. Overall, nearly 60% of the TGI people surveyed experienced some form of violence, threatening behavior, or harassment.
And for Black and brown trans women, whose experiences of transphobia may also coincide with misogynoir, racism, and anti-immigration rhetoric, they are at an even greater risk when it comes to experiencing violence and harassment.
Why is there an increase in violence against trans people?
When the webinar’s moderator, Senior Scholar of Public Policy Ayden Scheim, posed this question, Meyer pointed to the political “scapegoating” of trans people in the U.S. Under the current administration, there is a proliferation and mobilization of anti-LGBTQ+ hate that is especially rooted in anti-trans bias.
The higher numbers in recent data can also be explained by increased training for police when it comes to investigating and reporting anti-trans hate crimes and incidents. This could also be because more people are willing to report the violence they face.
Historically, though, TGI people report higher rates of distrust when it comes to seeking support from the police, so they often underreport the violence they experience. While researchers are working hard to collect a more “complete record” and a full portrait of anti-trans hate and violence, there are factors that can limit this work.
“Not everybody reports, not everybody who reports is assessed to actually be a hate crime, and not everybody who is assessed to be an actual hate crime is actually reported upward so that it gets into the data,” Meyer explained, detailing the difficulties researchers can face when trying to piece together a more “complete record” and full portrait of anti-trans hate and violence.
There is a “gap between legal protection and lived safety.”
Much of the distrust trans people experience when it comes to police and officials is a product of systems that have proven to be hostile towards trans people. “I feel like that tells us something that’s really important. Violence is not some random act…It is a pattern. It is structural. It interacts with social perceptions and economic vulnerability,” said Pamuela Halliwell, the Director of Behavioral Health Services at the San Diego LGBT Community Center.
Halliwell described her work as existing at the crossroads between behavioral health, community practice, and research, allowing her intimate insight into the tedious, chronic “hypervigilance” many trans people begin to embody as they face increased fear and stress from the threat of violence.
“It looks like people are being removed from their homes. It looks like fear, shame,” Halliwell described. “It looks like discrimination that feels overwhelming and contributes to a host of other mental health symptoms that become overwhelming and damaging. It looks like housing instability. It looks like communities are carrying communicative stress.”
While acknowledging that California has some of the strongest legal frameworks for trans people, Halliwell explained that there is a gap between stronger protections and the still prominent and tangible violence trans people face. She pointed towards a need for accessible survivor-centered reporting systems, making sure people know that these resources are available and ensuring that data collection and analysis moving forward really centers people across all gender identities.
How do we address our “structural vulnerability” and lean into different avenues of care?
Alec Watts, Assistant Deputy Director of Research and Policy at the California Civil Rights Department (CRD), explained that the department conducts extensive outreach to make sure communities and organizations are empowered with inclusive education. Part of this includes attending events as well as hosting trainings to educate people about civil rights protections, human trafficking, housing, and hate violence.
There are also direct ways members of the public can make their voice heard.
File a complaint
Watts explained that people are encouraged to file a complaint with the CRD directly if they believe their rights were violated. The department is in charge of investigating thousands of these complaints, and can provide mediation and, potentially, help file lawsuits in court on behalf of victims.
Consult in the CRD’s Community Conflict Resolution Unit
These are free, confidential resolution services intended to help community members when people are experiencing fear, conflict, or tension. Members of this unit can help facilitate discussion after an incident occurs, provide educational materials, and can assist schools in mediating tension between students, adults, or both.
Seek anonymous support through the California vs Hate hotline
The CRD operates the non-emergency reporting hotline for anyone in the state who has experienced or witnessed a hate crime or incident. But Watts stresses that this is more than just a reporting hotline — once you make a report, you are connected with a trauma-informed care coordinator who can connect you with legal, financial, mental health, or mediation resources. Services are free and available, regardless of gender, sexual orientation, race, or immigration status. Reports can be made at the website or on the phone, at 8338-NO-HATE.
“Our psyche cannot function in survival mode forever,” Halliwell said, pointing to a sustained fear that trans people navigate the world with. As severe and real as these fears are, Halliwell also clarified that TGI communities are not solely defined by harm. “The data [also] reflects a community that continues to show up, build networks, create chosen families, and demand better systems…It also highlights where intervention is possible. Structural vulnerability can be addressed, prevention is possible, and community care is real and expanding.”
Kristie Song is a California Local News Fellow placed with the Los Angeles Blade. The California Local News Fellowship is a state-funded initiative to support and strengthen local news reporting. Learn more about it at fellowships.journalism.berkeley.edu/cafellows.
-

 Hungary4 days ago
Hungary4 days agoHungarian authorities lift Budapest Pride ban
-

 Pride Special3 days ago
Pride Special3 days agoControversy looms as Kathy Hilton is named West Hollywood Pride Grand Marshal
-

 a&e features5 days ago
a&e features5 days agoWhy Michelle Visage needs you to get ‘PrEP Wise’
-

 Celebrity News2 days ago
Celebrity News2 days agoOutright International honors Cyndi Lauper at annual NYC gala
-

 Theater3 days ago
Theater3 days agoIn ‘A Man Called Mommy,’ Noadiah Eckman delivers queer joy across the aisle
-

 Colombia4 days ago
Colombia4 days agoClaudia López comes up short in Colombian presidential election
-

 Congress3 days ago
Congress3 days ago10 HIV/AIDS activists arrested on Capitol Hill
-

 a&e features5 days ago
a&e features5 days agoHow Saunder Choi crafts a queer anthem
-

 Politics4 days ago
Politics4 days agoFrom the desk of Equality California: The legal issues hitting California and beyond this Pride Month
-

 Federal Government3 days ago
Federal Government3 days agoAdvocates push back on proposed FCC warning labels










