AIDS and HIV
Lawsuit alleges manufacturer suppressing less kidney toxic AIDS drug
Suit alleges company concealed side-effects from patients and physicians
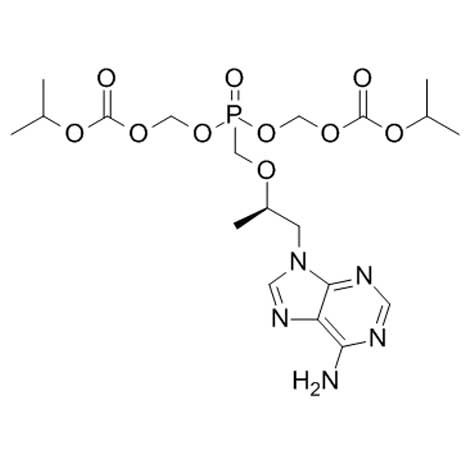

Lawyers for the AIDS Healthcare Foundation (AHF) filed personal injury and class action suits against Gilead on Tuesday, alleging the company stalled the development of a safer alternative to their highly-profitable HIV drug tenofovir disoproxil fumarate (TDF) for nearly two decades.
The personal injury action was filed on behalf of two Southern California men, both living with HIV, who claim they suffered bone and kidney damage from taking TDF at the doses prescribed. Behind the class action suit are two different Southern California men, who are also living with HIV and claim similar side effects from TDF.
Both complaints allege Gilead salespeople concealed from HIV-positive patients and their physicians information concerning the risks and side effects associated with TDF. They also claim the company declined to disclose the results of its studies, performed as early as 2001, which pointed to the viability and safety of their alternative formulation.
The Los Angeles Times reports Gilead has refused comment until the company can thoroughly review the complaints.
Several other widely-prescribed Gilead medications are made with TDF in combination with other HIV drugs. These include Atripla, Stribild, Complera, and Truvada.
In AHF’s class action, the organization’s lawyers seek to represent all California patients (1) who were prescribed Atripla, Truvada, and Viread (the brand name under which TDF was originally sold) from Oct. 26, 2001 through the present and (2) either personally, or via their prescribing physicians, were “exposed to Gilead’s misrepresentations.”
Regarding Truvada, among the claims presented in AHF’s class action is the charge that “Where Gilead did list potential patient concerns [with TDF], it misrepresented the risks as primarily for already-renally impaired or bone-compromised patients.”
According to the Los Angeles Times, the company paid doctors to conduct small clinical trials to test tenofovir alafenamide (TAF), the formulation Gilead scientists had found promising in their animal studies. The positive results from those clinical trials were shelved, according to the lawsuits, to protect the company’s TDF patents from competition and therefore drive up prices for the drugs.
Michael Lujano, one of the two plaintiffs in the personal injury suit, says he trusted his life to Gilead and the drug he believed would safely treat his HIV. “I’m bringing this lawsuit to try to hold Gilead responsible for their reckless focus on profits over patient safety,” he said.
The second plaintiff in AHF’s personal injury claim, Jonathan C. Gary, was diagnosed with a rare kidney disorder in 2010 and then with osteoporosis in 2017.
Attorneys for the men claim Gilead was twice warned by the Food and Drug Administration (FDA) concerning false claims by salespeople about the toxicity of TDF. The agency even recommended the company retrain its sales reps.
According to the company’s filings to the Securities and Exchange Commission (SEC), in 2017 Gilead earned $26.1 billion in revenue. Morningstar records of executive compensation indicate President/CEO John F. Milligan earned $15.4 million that year, while Executive Chairman John C. Martin earned $7.3 million.
AIDS and HIV
AIDS Healthcare Foundation announces 3 million people globally in its care
Los Angeles-based group lauded ‘historic milestone’

The AIDS Healthcare Foundation, a Los Angeles-based nonprofit group founded in 1987 that has become the world’s largest HIV/AIDS organization, has announced it has three million people in care around the world.
In a statement released on May 26, the organization, known worldwide as AHF, said the latest accomplishment reflects its global commitment to HIV prevention, care, and treatment. It says the accomplishment comes at a time when AHF marks the 25th anniversary of its first global programs launched in South Africa and Uganda in early 2001.
The statement says the three million people in care milestone also comes while the group approaches the 40th anniversary of its founding in 1987.
“Today, AHF provides lifesaving services in 50 countries across Africa, the Americas, Asia, and Europe, supporting millions of people living with HIV through a network of 1,056 global clinics, 79 healthcare centers in the U.S., 67 pharmacies, 96 wellness centers, 26 Out of the Closet thrift stores, outreach programs, and community partnerships,” the statement says.
“This accomplishment is far more than a number — it represents 3 million individuals whose lives have been touched by compassion, commitment, and the belief that healthcare is a human right,” Condessa M. Curley, the AHF board chair, said in a statement. “We extend our deepest gratitude to every member of the AHF team whose dedication made this milestone possible,” Curley said.
The AHF website notes the organization was founded in 1987 in Los Angeles as a network of hospices committed to “fighting for the living and caring for the dying” at a time when there was no effective treatment for HIV/AIDS. A statement on the website says since that time AHF has greatly expanded, converting its hospices into healthcare centers “and building a new paradigm for HIV care both in the United States and around the world.”
The statement adds, “Under the leadership of president and co-founder Michael Weinstein, AHF has grown from a group of friends dedicated to creating dignified hospice care to the largest AIDS organization in the world.” It says Weinstein “has been at the forefront of creating cutting-edge healthcare and advocacy programs and continues to drive the organization forward with the aim of saving more lives around the world.”
The statement announcing the milestone has also come at a time when more than 40 million people worldwide are living with HIV, “while hundreds of thousands continue to die annually from AIDS-related illnesses despite the availability of effective treatment.”
It says AHF’s response has included an expansion of its prevention and public health programs worldwide. In 2025 alone, according to the statement, AHF and its affiliated programs provided nearly five million free HIV tests globally and distributed more than 54 million free condoms, “underscoring the organization’s continued emphasis on both prevention and treatment.”
In D.C. AHF operates health care centers at 1701 K St., N.W., Ste. 400 [202-293-8680], 650 Pennsylvania Ave., S.E., Ste. 310 [202-350-5000], and 1647 Benning Road, N.E., Ste. 300 [202-350-5000].
AIDS and HIV
SB 1023 takes aim at the invisible insurance barriers to HIV prevention
While injectable PrEP represents a major leap forward in HIV prevention, outdated insurance systems are keeping it out of reach for the people who need it most.

Some bills pass through Sacramento with the kind of pizazz you’d likely expect from a Heidi Klum Halloween party, while others tip-toe it as quietly as a mouse. More often than not, it’s the latter that carries more weight. SB 1023, recently advanced by the Senate Appropriations Committee and authored by Senator John Laird, falls into this second category. On paper, it’s about insurance billing codes and reimbursement pathways for injectable PrEP. In real life, it’s about whether prevention is actually accessible, or just theoretically available if you know the right pharmacy, the right paperwork, and the right brand of patience.
Let’s put this one in real talk. Injectable PrEP is one of the most promising tools we have in regard to HIV prevention at the moment. It’s long-acting, clinically effective, and designed to reduce the hassle of daily adherence that comes with its pill-form predecessor. The issue SB 1023 is looking at is not medical but bureaucratic. Some health plans cover injectable PrEP, but then funnel it through pharmacy benefits that don’t actually work for outpatient clinics trying to actually administer it. The result is a kind of administrative purgatory. The medication is there, the science is too, the patients are eligible and ready to go, and yet access gets stuck in the pipelines of insurance systems that were never designed for prevention as care. It appears that, when it comes to real care, they simply don’t.
Now, more than ever, even with the oral daily PrEP that we all know, love, and pop, this matters because prevention only works when it is accessible to the public that needs it, policy documents aside. When talking about PrEP and HIV prevention, we fall into the formulaic thinking of pop a pill, reduce the risk, and avoid the worst. Period. But anyone who has actually lived inside this system knows it’s more complicated than that. Prevention is impacted by income, stigma, clinic hours, insurance literacy, access to pharmacy, and whether you feel comfortable enough in a medical setting to keep trying. It’s shaped by whether your provider understands queer health without making you feel like a statistic instead of a person.
And for LGBTQ+ communities (especially queer and trans folks, Black and Brown communities, and sex-working communities), “usable” is the whole game. At face value, this is a policy fix. Realistically, it’s the recognition that HIV prevention still doesn’t distribute itself evenly across populations or communities.
There’s also something psychologically significant that’s happening here. Prevention is an emotional issue just as much as it is a medical issue. When access is accessible and stigma-free, folks can actually plan their lives without constantly negotiating fear and apprehension along the way. When unaddressed, you get something else entirely. Anxiety dressed up as responsibility. That specific flavor of anxiety that says, “You should be doing more,” while quietly making it harder to get up, get out, and do anything worthwhile.
I‘d be hard-pressed to harp on this topic without speaking on my own experience. I take Descovy every day. Not out of simple mindless routine, but as a practice of self-care. Like brushing my teeth, except with a much heavier historical and cultural context behind it. Some days it feels empowering, while other days it feels more like a reminder that prevention in queer life has never at all been passive. It is something we actively maintain in a world that has not always been necessarily interested in maintaining us.
And occasionally (and I say this with no shame), I also use doxycycline as I see fit in the context of post-exposure prevention practices. That, too, is part of a much broader shift in how we think about HIV prevention. It is less about singular solutions and more about layered strategies that meet people where they actually are, not where public health PowerPoints imagine them to be.
But the most real part about all of this is that, even when you are doing everything “right,” there is still an underlying buzz of vigilance. The medical history doesn’t lie. The knowledge that access can always tighten, loosen, or straight up disappear depending on politics, funding, or the latest moral panic.
That’s why SB 1023 matters. More than injectable PrEP, it’s about whether the systems around HIV prevention are evolving toward coherence or continuing to rely on the assumption that individual folks will simply absorb the friction. For our queer community, this bill sits inside a larger tension. We are simultaneously in an era of unprecedented biomedical capability and persistent structural inequity. We can prevent HIV with remarkable effectiveness, yet still struggle to make prevention consistently available across all the places it needs to be.
This gap has consequences for both physical and mental health. Physically, inconsistent access means preventable infections still occur. This is not only because tools don’t exist but because systems don’t deliver them cleanly. Mentally, it produces a quieter, more chronic strain: the stress of managing prevention as a personal project rather than a collective guarantee. It’s the difference between “I am protected” and “I hope I’ve done enough.”
This particular difference lives in the body. It shows up as vigilance fatigue. As decision exhaustion. As the low-level cognitive load of always tracking when prescriptions need refilling, when appointments need scheduling, and when insurance requires prior authorization that no one warned you about until you were already at the pharmacy window. And for communities already navigating stigma in healthcare settings, that burden is not evenly distributed.
So what do we do with a bill like SB 1023? On an individual level, we continue to use the tools and resources available to us, and we talk about them without shame. We normalize prevention not as a moral achievement, but as routine care. We share information in ways that don’t assume everyone has equal access to providers who understand queer health without hesitation or bias. We check in on each other not just about risk, but about access: “Are you actually able to get what you were prescribed?” We also resist the idea that prevention should feel isolating. No one should have to individually solve what is fundamentally a systems problem.
On a macro level, SB 1023 is a reminder that policy details are not details at all, aside from the difference between access and obstruction. It points toward a far more reaching need for healthcare systems designed around continuity rather than fragmentation. Systems that don’t require patients and clinics to constantly translate between medical intention and insurance interpretation.
It also raises the bigger question: if we already know how to prevent HIV, what exactly are we waiting for to make prevention universally frictionless? The science is not lagging. The barrier is administrative will. And administrative will is, in its own way, a form of public health intervention.
If we get it right, prevention becomes quieter. Not invisible, but integrated. Something you don’t have to fight for every month. Something that doesn’t require you to constantly re-prove your eligibility for safety. If we get it wrong, we continue to rely on individuals absorbing the inefficiencies of systems that were never built with us in mind.
I don’t think this bill is the end of anything. But it is a signal, small, specific, and important, that the conversation is shifting from “Do we have the tools?” to “Why are the tools still so hard to reach?”
For those of us who live inside that gap between availability and access, that shift is not in the least bit abstract. It’s very real, very personal, and long overdue. But that’s just one very gay man’s opinion.
AIDS and HIV
Fearless in the face of financial cuts: Alex Garner on HIV advocacy in 2026
As HIV programs face devastating cuts worldwide, Alex Garner explains why visibility, pleasure, and resistance remain acts of survival

At a time when global HIV funding is being absolutely gutted, and anti-LGBTQ rhetoric is at the mic 24/7, Alex Garner has little to no interest in softening the truth. As a longtime activist and leader at Mpact Global, Garner speaks with the sense of urgency that can only come from lived experience and witnessing history threaten to repeat itself in real time. For Garner, these financial cuts are not some abstract policy debate or a budget footnote. They are, with no doubt, life-and-death decisions that disproportionately endanger our queer and migrant communities and everyone living with HIV around the world.
In our conversation with this beacon of hope, Garner reflects on surviving three decades with HIV, the groundbreaking changes in queer sexual health since the early years of the epidemic, and why today’s political climate needs bold voices rather than shy whispers. As fearless as he is deeply compassionate, Garner makes the case that modern HIV advocacy is about pleasure, dignity, visibility, and refusing to let queer lives be treated as expendable. just as much as it is about medical treatment. PREACH.
Around the world, we are seeing funding for HIV and AIDS programs slashed. What is the undeniable and very real impact of these cuts, especially for communities of color?
Funding numbers may seem like figures on a spreadsheet to some, but they represent real lives. Queer people have and will die. That’s it in the most basic terms. Around the globe, queer people are severely vulnerable and are already working hard to get resources to support the community. Migrants, people living with HIV, and gender diverse folks are going to bear the brunt of these cuts.
What makes right now such a critical turning point for global HIV advocacy?
The science of HIV has made incredible advancements. We have the tools to radically change this epidemic, but we are lacking the will from those in power. They simply don’t care enough about our lives to make an investment. It has been 45 years since the HIV epidemic, and Mpact and our partners are still fighting for basic resources. Each day, we must continue to demonstrate that our lives have value and meaning.
Testing and education are often the first to go when budgets cut. Why are those two areas so essential to maintaining much needed progress?
Education is fundamental to public health. 45 years into the epidemic, we know that when it comes to community education, unfortunately, we have to do it ourselves. Mpact has worked in collaboration with various partners and social media influencers around the globe to create health promotion materials and content because it is needed now more than ever. Working with the community to ensure that health education is understandable, relevant, and culturally appropriate is critical to effective education initiatives.
Mpact Global is choosing to expand at a time when many organizations are pulling back. What’s behind that decision?
The anti-LGBT movements are growing around the world, and they expect us to pull back, but we are stepping forward. We must demonstrate that we are not afraid, that we are not going anywhere, and that the lives of our queer community matter and are worth fighting for. We also have a responsibility to step up since there are many partners around the world in places like Senegal, Ghana, or Indonesia, who are unable to do so without immense risk. We have to use our power and voice for them and the very real issues they face.
As someone openly living with HIV, how does your own story impact the way that you approach this work?
I have been living with HIV for 30 years. I have been HIV-positive for more years than I was HIV-negative. It impacts every aspect of my life. It’s created resilience, resistance, and a drive to keep fighting. Stigma is not going to hold us back, and we have to approach our work with a sense of fearlessness and shamelessness. We can build a community for queer people living with HIV, as it can empower people living with HIV while at the same time allow negative people to witness that a community is possible and that living with HIV is not something to be scared of.
Since the start of the 2000s, how has the world of queer health advocacy changed and what has gone unchanged?
In the past 26 years, the world of HIV has drastically changed. We understand that with successful treatment, people living with HIV can achieve an undetectable viral load. The primary benefit of being undetectable is that you can live a long and normal life. The secondary benefit is that it is impossible to transmit HIV. PrEP is also more than 10 years old. Both these advancements have changed the quality of life of gay men and have changed our sex lives.
We’ve witnessed a mini-sexual revolution where more gay men are pursuing the sex they want. They have prioritized pleasure and are not living in fear and anxiety due to HIV. Our desire for pleasurable sex and for connections has not changed, but the world around us has. We have dating apps and social media, for good and for bad, and we have Doxy PEP. It’s one more thing to allow gay men to have sexuality that is centered on pleasure and sexual satisfaction and not eclipsed by fear and risk. While many gay men may not have experienced the 80s and 90s, we don’t want gay men to live in fear.
With political hostility and legislative attacks are on the rise, how are HIV prevention and care efforts affected?
During these hostile times, anything that is seen as promoting the sexuality of gay men is the first to get cut. We’ve had similar things happen around reproductive health and health for migrants. It’s all interconnected, and it’s from a familiar playbook. It requires resolve, creativity, and coalition building to endure these attacks.
With Mpact Global operating in 62 countries, what are the common threads you’re seeing across regions?
Over 60 countries still criminalize same sex sexual behavior. Meanwhile, governments seek to criminalize us because of our HIV status, gender expression, sex work, or migration status. Responding to laws, policies, and authoritarian leaders who criminalize us is a through line. We mobilize, and we strategize. And at the same time, we find and build community, and we continue to raise our voices and put forth a narrative that demonstrates our humanity and need for fundamental human rights.
What does effective HIV advocacy look like today, as opposed to 3 decades ago?
Education, access, and control over one’s body. That has not changed much, and those are things central to HIV, reproductive justice, and basic human rights.
For younger generations who didn’t live through the early years of the epidemic, how do you convey the relevance and gravity of the issue?
We lived through the dark and difficult times so that the next generations don’t have to. I never want gay men to have to live in fear, shame, and guilt. I want them to be able to pursue their sexuality with abandon, passion, and desire.
We need to show them that our lives matter, that we are continuing to fight for our health and our lives all around the world, and that they need to be a part of it. We’ve got to work together. I want them to embrace their sexuality and never take it for granted. To not feel ashamed about their sex but to explore, and grow, and find pleasure.
How does intersectionality impact the future of HIV advocacy?
Everything we do is political, and all parts of our lives intersect with a variety of issues. Recently, the Mpact the Besoton Sidoso Internacional or International HIV Kiss-In was a public activation on the border in Tijuana to demonstrate the sexuality of queer people living with HIV and to highlight the issues faced by migrants on both sides of the border in relation to health, HIV, and sexuality.
What role has resilience and adaptability played in the work that you do with Mpact?
Mpact’s 20 years are defined by resilience. We’ve had to continue to fight for health and human rights even amidst very challenging times. There have been successes, but it’s evident, now more than ever, that gains, like access to antiretroviral medication, must continue to be fought for as they are not guaranteed. And even things we expect to be guaranteed, such as human rights, continue to be at risk, so we must be nimble in our response and organizing.
Where do you see HIV and sexual health advocacy in the next decade?
We need to normalize sexual health as part of our culture. They are viruses and bacteria and nothing to be ashamed of. We need to integrate them into our overall wellness, just like we do with cold and flu season. That requires more than just traditional advocacy; it requires a cultural shift. Culture will change and policy will catch up, just like with marriage equality.
What innovations, whether political, medical, or communal, give you a taste of hope at our current moment?
Social media is still an underutilized resource, particularly by progressives. Seeing queer people all around the world leverage social media to speak openly, honestly, and unashamedly about who they are and what they experience can transform our world and our politics. When queer people openly express themselves amidst many risks, it’s always a source of hope and inspiration.
If you could speak directly to global leaders and those responsible for slashing budgets, what would you want them to understand about the work that you do?
They need to understand that real lives are at risk. But sadly, I think that does not move them. People are dying and suffering all over the world, and policymakers are doing nothing. If they can’t lead, respect, and advance human rights, then they are not leaders, and they need to give up power and let others do the work.
As Mpact Global celebrates 20 years, what legacy do you hope the next 20 years will build for the next generation?
Community building is central to the work we do. We want to be a force for good that strengthens queer community and works in collaboration to ensure people can freely express their sexuality and gender, have control over their bodies, and continue to fight for health and human rights. Community has always been the legacy for queer folks, and we are dedicated to continuing that part of our experience.
Follow all things MPact Global
AIDS and HIV
Congresswoman Maxine Waters introduces new resolution for National Black HIV/AIDS Awareness Day
H.Res.1039 supports more funding, resources and awareness for Black American communities, who are disproportionately impacted by HIV/AIDS.

Today is National Black HIV/AIDS Awareness Day. Advocates established this day of awareness on Feb. 7, 1999, and nearly 30 years later, Black communities in the U.S. continue to be disproportionately impacted by HIV.
On Wednesday, California Congresswoman Maxine Waters introduced H.Res.1039, a resolution that supports the goals of National Black HIV/AIDS Awareness Day and calls for a collective commitment to address disparities Black people with HIV face. Waters represents the state’s 43rd congressional district, a majority Black and Brown population comprising South L.A. cities like Hawthorne, Gardena, and Inglewood.
In the resolution, Waters urges state and local government officials, as well as their public health agencies, to acknowledge the importance of this awareness day and encourage their constituents to get tested for HIV. The resolution also requests that the Secretary of Health and Human Services prioritize distributing grant funding to minority-led, HIV organizations and community-based approaches to fighting HIV stigma, LGBTQ+ discrimination, and racism.
In 2023, young Black men accounted for 47% of new HIV diagnoses among youth, while young white men made up 3% of these diagnoses, according to a new Williams Institute report. Black women also have the highest HIV diagnosis rate among women, and Black community members overall represent 38% of new HIV diagnoses and 39% of people living with HIV in the U.S., despite being only 12% of the national population.
Beyond the disproportionate rates of infection and diagnosis amongst Black Americans, these communities also face greater difficulties in accessing the medical care needed to prevent and treat HIV. In the same year, white Americans were 7 times more likely to access Pre-exposure Prophylaxis (PrEP) compared to Black Americans, a data point that affirms racial and healthcare inequities Black people continue to face in the U.S.
“[This] is a day to commemorate the impact of HIV/AIDS on Black Americans and encourage continued efforts to reduce the incidence of HIV, eliminate health disparities, improve access to care and treatment, and show support for all those who are living with HIV/AIDS,” said Congresswoman Waters, in a press release.
Waters has been an advocate for people impacted by HIV/AIDS since the peak of the crisis in the 1980’s. In 1998, she worked to establish the Minority AIDS Initiative, which expanded national prevention and treatment efforts in support of minority communities, who remain disproportionately impacted by HIV. In 2025, Waters introduced the “HIV Prevention Now Act” as well as the “PrEP and PEP are Prevention Act,” to increase prevention efforts and reduce health insurance barriers to access preventative resources, respectively.
H.Res.1039 is the latest addition to the congresswoman’s efforts to raise awareness for Black and other minority communities impacted by HIV/AIDS, and to fund and support on-the-ground efforts that prioritize their care and wellbeing.
The resolution is endorsed by various LGBTQ+ organizations mobilizing for communities impacted by HIV, including AMAAD Institute (Arming Minorities Against Addiction and Disease), LA Pride, AIDS Foundation Chicago, and PFLAG National. The resolution is also co-sponsored by 29 other U.S. representatives, including fellow California congressmembers Robert Garcia, Laura Friedman, Nanette Barragán, Sydney Kamlager-Dove, Lateefah Simon and Mark Takano.
H.Res. 1039 has been referred to the House Committee on Energy and Commerce, and currently awaits further action.
Kristie Song is a California Local News Fellow placed with the Los Angeles Blade. The California Local News Fellowship is a state-funded initiative to support and strengthen local news reporting. Learn more about it at fellowships.journalism.berkeley.edu/cafellows.
AIDS and HIV
From ACT UP to apps: A candid conversation with MISTR founder Tristan Schukraft
As HIV prevention ushers in a new era, Schukraft reflects on the evolution of sexual healthcare and the power of stigma-free prevention

It was not too long ago that an HIV diagnosis was read as a death sentence. In its earlier decades, the HIV/AIDS crisis was synonymous with fear and loss, steeped in stigma. Over recent years, open conversation and science have come together to combat this stigma while proactively paving the way for life-saving treatments and preventive measures like PrEP. Now, in 2026, with discreet and modern platforms that meet people where they’re at in their lives, HIV prevention has evolved from hushed words of warning into something far more sex-positive and accessible. Game-changing services like MISTR are a testament to this shift, showing our community that healthcare doesn’t have to feel clinical or shaming to work. It can be empowering and, dare I say, celebratory.
Few people embody this evolution quite like Tristan Schukraft, founder of MISTR. With one hand in healthcare and the other high-fiving through queer nightlife, Schukraft gets that, from the bar to the bedroom and beyond, prevention happens in person and in real life. His approach has helped turn PrEP, DoxyPEP, and testing into normalized parts of our daily queer life, reaching hundreds of thousands of people across the US.
In our conversation, Schukraft shares candidly about stigma, policy, and why the future of sexual health depends on keeping it real.
You have one hand in healthcare and the other in nightlife and queer spaces. Can you share with us how these two spheres impact and inform each other? How do they impact and inform you?
Honestly, for me, they’ve never been separate. Nightlife and queer spaces are where people meet, date, hook up, fall in love, and make friends. That’s real life. Being in queer spaces all the time keeps me grounded and reminds me who we’re building MISTR for.
MISTR markets sexual health in a sex-positive, stigma-free fashion. Can you share with us how you measure the impact of this approach?
This year, we held the first-ever National PrEP Day. Dua Lipa performed, and Cardi B was there. After the event, Cardi B went on her Instagram live to encourage people to sign up for PrEP.
When you make sexual health stigma-free and sex positive, people talk about it. We see it in how people use the platform. When 700,000 people are willing to sign up, get tested, start PrEP, and add things like DoxyPEP, that tells us we’ve made it feel safe and normal instead of scary or awkward. And then we see it in the results. Since we expanded DoxyPEP, STI positivity among our patients dropped by half.
How have you seen the conversation of sexual health in our LGBTQ+ community change in mainstream culture in recent years?
Ten years ago, nobody was casually talking about PrEP, and if they did, it likely referenced one being a Truvada whore. Now it’s part of the culture. Popstars like Troye Sivan post pictures of their daily PrEP pill on social media. Cardi B goes on Instagram Live telling people to get on PrEP.
For many sexually active gay men, taking PrEP is simply part of the gay experience. For people in more remote areas, it might not be as talked about. Particularly in rural or more conservative places, MISTR can be a life-changing option. No awkward visits to the family doctor or the local pharmacy where everybody knows your business. It’s all done discreetly online and shipped straight to your door.
You have publicly argued that cuts to government HIV prevention funding are of high risk. Would you please elaborate for us on what those budget decisions mean on an individual level?
It means real people fall through the cracks. Someone doesn’t get tested. Someone waits too long to start PrEP. Someone finds out they’re HIV-positive later than they should have. Community clinics will be the hardest hit, especially those in underserved communities. The good news is that MISTR is ready to help people who might lose their access to care. All you need to do is sign up at mistr.com, and it’s totally free with or without insurance.
From your (and MISTR’s) perspective, how do these funding cuts threaten ongoing efforts to end the HIV epidemic?
For the first time, we have all the tools to end HIV. If everybody who is HIV negative is taking PrEP and everyone HIV+ is virally suppressed, we can end all new HIV transmissions in the United States. We have everything we need today. All we need is to get more people on PrEP. Cutting funding risks losing that momentum. Ending HIV requires scale and consistency. Every time funding gets cut, you lose momentum, trust, and infrastructure, and rebuilding that takes years.
HIV transmissions don’t pause because budgets change.
In our current climate of decreased federal investment, what role do you feel private healthcare and business should play in sexual health?
With reports that the current administration is considering cuts to HIV and prevention funding, we face a moment of reckoning. At the same time, some employers are seeking to exclude PrEP and HIV prevention from their coverage on religious freedom grounds. If these challenges succeed, and if federal funding is slashed, the consequences for public health will be devastating. But this is where the private sector must step up to fill the gap, bridge divides, and deliver results.
Businesses have the power and platform to normalize HIV prevention and drive measurable outcomes. At MISTR, we see firsthand what’s possible: since introducing DoxyPEP, STI positivity rates among our patients have been cut in half. But it’s not just about medication. It’s about messaging.
Our sex-positive, stigma-free marketing speaks directly to our community, making sexual health part of everyday life. No awkward doctor visits, no needles, no paperwork — just free online PrEP and STI testing, prescribed by real physicians and delivered to your door. That kind of impact could grow exponentially if more employers embraced this approach and made HIV prevention part of their employee wellness programs.
Employers, this is your call to action. Start by making sure your health plans cover PrEP and DoxyPEP. Partner with platforms like MISTR to give employees private, stigma-free access to care. Offer on-site testing. Talk openly about sexual health, not just during Pride, but every day of the year. This is not political — this is about protecting lives, strengthening communities, and building a healthier, more productive workforce. Because healthy employees aren’t just good for public health — they’re good for business.
When the private sector steps up, outcomes improve. And when businesses align with platforms like MISTR, scaling impact isn’t just possible — it’s happening.
Has MISTR experienced any direct effects from these recent shifts in public health funding?
MISTR’s unique model is totally free for patients with or without insurance, and we don’t cost the government or taxpayers a penny. We are scaling up our efforts to reach people who might be losing their access or care.
What would be your message to policymakers who are considering further cuts to HIV/AIDS programs?
During his first term, President Donald Trump committed unprecedented resources to the Ending the HIV Epidemic initiative here at home. Bipartisan support has shown what’s possible when bold leadership meets smart strategy. To policymakers: I urge you to reconsider any cuts to HIV prevention funding. This is not the time to pull back. It’s the time to push forward. Ending HIV is within reach — but only if government, private industry, and community organizations stand together.
What is one perhaps overlooked win from last year that impacted you on a personal level?
Seeing our STI positivity rate drop by half after expanding DoxyPEP.
Looking at the year ahead, what are MISTR’s most significant priorities for sexual health in 2026?
Expanding access, especially in the South and in communities that still get left out. Rolling out injectable PrEP. And just continuing to make sexual healthcare easier and more normal.
Is MISTR planning to integrate injectable PrEP into the mix?
Yes, absolutely. Long-acting PrEP is a game-changer, especially for people who struggle with daily pills. We’re rolling it out through a hybrid model: telehealth plus in-person injections in key LGBTQ+ neighborhoods.
As you just mentioned, MISTR reported cutting STI positivity rates in half through expanded DoxyPEP access last year (mad props). What do you think allowed you to accomplish this?
We made it easy, normalized it, and bundled it into care. No stigma, no hoops, no lectures. When you remove friction, people take care of themselves.
If you could eliminate from our collective consciousness one particular myth or stigma about sexual health, what would it be?
Sexual health should be talked about in the open, and we can have fun with it! I threw a party in October for National PrEP Day. How many parties have you been to celebrating sexual health? You should go to more.
What were the most significant obstacles or setbacks you saw some of your patients face in accessing PrEP and STI prevention services in the recent past?
Stigma, paperwork, insurance, provider judgment, and just navigating a system that was never designed for them.
Looking at the next chapter, what would you like to see MISTR accomplish next? And I have to ask, what’s next for you?
For MISTR: scale, injectables, and getting more HIV+ utilizing MISTR long-term HIV care. I started MISTR with the goal of ending HIV, so this year, next year, and as long as it takes, I want to make history and end HIV.
For me: staying close to the community, building things that matter, and making sure we never lose sight of the humans behind the metrics.
For more information, head to HeyMistr.com.
This article is part of LA Blade’s January health series.
AIDS and HIV
Community is the cure: AIDS Walk LA returns to fight HIV and funding cuts
AIDS Walk Los Angeles returns to West Hollywood on October 12 with the theme ‘Community Is the Cure,’ highlighting the vital role of unity, radical community action, and advocacy in the fight against HIV/AIDS, stigma, and government funding cuts

APLA Health, a nonprofit providing HIV care, prevention, and sexual health services, announced the return of AIDS Walk Los Angeles on Sunday, October 12, 2025, starting from West Hollywood Park. This year’s theme, “Community Is the Cure,” emphasizes the role of unity in advancing progress against HIV/AIDS and supporting those affected.
Walk day will feature a live performance from RuPaul’s Drag Race star Heidi N Closet, DJ sets, and community booths. Following the celebration, walkers will make their way through the streets of West Hollywood.
“This event was born out of urgency, and it’s just as relevant today,” said Craig E. Thompson, CEO of APLA Health. “We’ve made incredible progress in the fight against HIV, but that progress is under direct threat from funding cuts and political attacks. Now is the time to show that we won’t be silenced or set back.”
Since its inception forty years ago, AIDS Walk Los Angeles has grown into one of the largest HIV/AIDS fundraising events in the world, raising nearly $100 million. Proceeds fund APLA Health’s services, including HIV specialty care, sexual health services, food and nutrition support, and housing assistance.
“The HIV/AIDS epidemic has always shown us that progress happens when people come together, when patients, neighbors, families, activists, and health providers stand shoulder to shoulder. Today, with funding under attack and stigma resurfacing, unity is not optional. ‘Community Is the Cure’ is a reminder that science alone isn’t enough. We need collective willpower, advocacy, and solidarity to ensure everyone has access to the care and dignity they deserve,” Thompson said.
Cuts to Medicaid, the Ryan White Program, and prevention funding threaten access to medications, housing assistance, and food security. “Here in Los Angeles, where tens of thousands rely on these programs, even modest reductions can push people back into crisis. AIDS Walk Los Angeles helps fill some of those gaps, but philanthropy cannot fully replace the government’s responsibility,” Thompson explained.
Communities of color, the medically underserved, people living in poverty, and those experiencing homelessness are disproportionately affected. “The barriers aren’t medical. They’re structural: stigma, lack of insurance, unstable housing, and underfunded safety-net services. The administration is underinvesting in the very programs designed to break down these barriers. Until that changes, inequities in HIV prevention and care will persist,” he said.
Funds raised by the walk provide housing, groceries, case management, and access to medical care. Thompson shared the story of one patient who had lost housing and had to choose between medication and meals. “Through AIDS Walk–funded programs, we were able to connect him to stable housing, consistent medical care, and our food pantry. Today, he is virally suppressed, working again, and mentoring others who are newly diagnosed with HIV. Stories like his are common, and they remind us that cuts aren’t just numbers on a spreadsheet, they’re setbacks in real people’s lives.”
Beyond fundraising, AIDS Walk LA is a platform for advocacy. “When thousands of people flood the streets of West Hollywood, it’s a visible reminder to our community that HIV/AIDS has not gone away and to policymakers that their decisions have life-or-death consequences. The walk puts pressure on elected officials to fund programs, fight stigma, and stand up for people affected by HIV,” Thompson said.
He urged the public to take action beyond walking and donating. “Call your elected officials. Demand that HIV services remain fully funded. Show up for community hearings. Support housing initiatives. Share facts on social media to counter stigma. And, importantly, talk. Without continued conversation about HIV/AIDS and sexual health, we jeopardize the incredible advancements we’ve made in HIV care and prevention.”
Thompson also framed the walk as part of a broader activist approach. “Radical action today means showing up for one another in tangible ways, housing someone, feeding someone, advocating for someone, and refusing to accept policies that erase people’s humanity. As our theme emphasizes, when politicians fail us, COMMUNITY IS THE CURE. For participants in AIDS Walk LA, it means recognizing that the walk is just the beginning. Each person can be a messenger, an advocate, and an ally in daily life. When we work together to get people connected to support services, the collective action is radical because it insists on care and dignity in a time when those values are under attack.”
“Activism ensures policymakers cannot quietly dismantle programs. Community solidarity means that no one gets left behind. Public pressure creates accountability. When thousands unite at AIDS Walk LA, it demonstrates the broad mandate to keep fighting until HIV is no longer a public health crisis. That unity is as important as any medical breakthrough,” Thompson concluded.
For more information on AIDS Walk Los Angeles and how to register, visit https://AIDSWALK.LA.
AIDS and HIV
“If not now, when?” Journalist and activist Karl Schmid is reshaping how we talk about HIV/AIDS
The Blade sits down with the award-winning host to talk about stigma and his latest initiative – taking HIV education onto the runway

Karl Schmid has long been a staple in queer news and media. Hailing from Australia, Schmid grew accustomed to the limelight from a young age, and eventually made a successful transition into American network television through his work on red carpets for ABC.
In 2018, he would be thrust into the spotlight for a different reason. Despite repeated warnings from professional acquaintances, Schmid came out as HIV positive to share his story, wear down stigmas, and to educate people about important and misunderstood science like the concept of undetectable equals untransmittable.
And so, Schmid would grow accustomed to renewal. In 2019, he launched Plus Life Media, a platform that shares HIV resources, education, and stories of resilience from people living with HIV/AIDS. He was part of the team behind the Emmy-nominated short documentary, Marty’s Place: Where Hope Lives: an intimate look into a housing cooperative created in the 1990s to support people with HIV/AIDS. Late last week, he flew to New York for the U.S. debut of his latest initiative: HIV Unwrapped. The project pairs researchers and scientists in HIV studies with fashion design students, culminating in a runway show that features bold reimaginings and interpretations of crucial science.
The Blade spoke with Karl about the cross-pollination in his own life and career, and how his latest effort is shaping the way he blends activism and media to reinvigorate conversations around HIV/AIDS.
HIV Unwrapped feels like a really interesting pivot into a different kind of modality and platform for you. Can you tell me more about the initiative and how it fits into your legacy of activism and work?
Well, HIV Unwrapped came out of Australia and this wonderful activist named Brent Allan, who I’m good friends with. And Brent had been at, I think, the WorldPride, or something that was happening in Australia, and there was an exhibition where they paired some engineers and science people of varying professions with a fashion designer to try and illustrate their work.
And he thought, “Wow, what a great way to maybe try and interpret HIV science and reignite the conversation around HIV.” So it started in Melbourne, Australia, and it was successful there. And then they did a version in the UK earlier this year. And then in July for the International AIDS Conference that took place in Kigali, in Rwanda, they did a version.
And that’s kind of where I stepped in. I think with HIV Unwrapped, you’re drawn into the fashion first — and then the science comes. It’s just a really unique and different way of reframing the conversation about HIV. But beyond that, it’s also working with fashion students, young people, and to see them become so invested in HIV science. That’s what gets me the most excited. Because you’re opening a whole new world to a person who doesn’t maybe really think about HIV all that much, and now they’ve learned all this amazing stuff. Suddenly you’ve got a whole new bunch of people talking about HIV in exciting and fresh ways, and they’re telling their friends and they’re having conversations about it.
You’re trying to create different entry points for people to not only talk about the science behind HIV and AIDS, but also how we can reinterpret it in original and visually interesting ways.
I mean, look, you’ve got the House Appropriations Committee willing to advance this “Big Beautiful Bill” that slashes $2 billion in HIV funding in this country, and as someone living with HIV, you keep going, “Why would they do that? Why? We are so close to ending HIV.” All of a sudden, we’ve stopped funding research, and we’re stopping this, and I’m just like, “Why?”
Now more than ever, we’ve got people and politicians and government and a large part of the United States who think, “Oh yeah, HIV isn’t a thing anymore. It’s not important.” Well, it is still a thing. It is still important, and HIV Unwrapped is a way to be creative and open new avenues of dialogue.
A friend of mine sent me a handwritten note the other day. This is in Texas. One of his roommates wrote a handwritten note to another roommate, saying, “Oh, just so you know, ‘such and such’ has AIDS. So I’d be scrubbing the shower if I were you after he uses it. I personally have decided to go and shower at the gym.” And this is a person in their 30s, so we’re still having these dumb conversations from 1984.
That’s why I do what I do with Plus Life, and that’s why I’m like, “Wow, HIV Unwrapped is another tool in our toolbox we can use.” And I guess our hope is really that scientists will see this across the country and around the world and go, “Well, that is a really cool way to explain my science and make it accessible,” and want to engage in future projects like this.
How queer communities are represented in the media can leave deep imprints. I was watching an older interview you did where you recall this HIV ad you saw in a movie theater as a kid. The Grim Reaper’s there, and it’s creepy and sinister. It seems like we’ve moved away from that kind of imagery — but it doesn’t feel like we’ve necessarily come to a much better understanding of HIV/AIDS. Where do you think we’re at now in terms of representation?
The fact that, to this day, I can tell you where I was, what the movie was, that it was a Saturday, which cinema it was in, and I was a seven year old kid. It gives you an idea of how burned into my memory that is. And, I agree. I think we’ve moved away largely from the fear tactics, but we’ve stopped talking about it. I’d like to think that we’ve gotten better in representing people living with HIV, on television and film, as just people living with HIV. Where HIV doesn’t become the central storyline, and it’s not a doom and gloom, “woe is me” story. It’s just part of who they are.
But we sort of dance around anything that’s to do with our bodies or sex or sexuality, because it makes us uncomfortable. And so when I pitch stories to ABC and other networks, and even, quite frankly, trying to get press to cover HIV Unwrapped: we get passed on. People don’t want to talk to us because HIV isn’t sexy. As long as we keep doing that and having that reaction — if we don’t talk about it — we don’t get to a place where we can be comfortable with it.
Since I came out about my status all those years ago, my focus has been trying to normalize the conversation. That’s how we grow, and that’s how we learn — whether it’s HIV, whether it’s politics, whether it’s anything. But if we don’t talk about it, and we sort of pretend it doesn’t exist, and then we cut funding for it, and we sort of sweep it under the rug: it ain’t going to go anywhere.
Women of color, Black and brown women, have the highest infection rates in the United States. In the south those rates keep going up and up and up, and now we’re cutting funding to testing and to counseling and to services. I’m really worried that we will end up back with AIDS wards in hospitals again, and there’s no need for it.
To bring it back to HIV Unwrapped, that’s why, if I can come up and work with an amazing group of people to tell stories and to have conversations in fresh ways, then I’m going to keep doing it — whether it’s HIV Unwrapped, whether it’s what I do on Plus Life, whether it’s a television show or whatever crazy thing I come up with next.
It feels like there’s so much watering-down in terms of conversations around sexual health, HIV and AIDS. What is the resistance that you’ve faced, and how has that resistance changed over the years?
I’ve heard, “I think what you’re doing is great. It’s just not the right time to tell the story.” Well, when is the right time to tell the story? I think so many people in America think HIV doesn’t affect them. And the reality is very different. HIV affects everybody. If not now, when? People are still dying in this country. People are afraid to get tested, and they’re living in shame. And they’re hiding what’s wrong with them because of the stigma — and that just shouldn’t be happening.
I’ve said this a million times too. You know, HIV and AIDS doesn’t kill you. That’s not what’s going to put me in the grave. But it’s the stigma. It’s the bigoted, out-of-date opinions of Congress and politicians and public figures and uneducated people who just think this is somehow some deviant, dirty “sex disease” or drug disease, and those of us who get it deserve it. And you know, television networks, especially, are in the business of making money. And to do that, they sell advertising, and so they’re worried that advertisers will turn off if you talk about this kind of stuff.
And I think it’s maybe a little bit the opposite. I think if you can find, again, engaging and interesting ways to have real conversations, I’ve certainly found that you get a very vast and wide audience of people who will chime in and may have an opinion. And again, our opinions may differ, and that’s okay, but, but as long as we’re having the conversation, the more you say it, the less scary it is.
Almost 20 years after you were first diagnosed with HIV, do you see yourself pivoting now to work more on activism? Is that something you’re trying to focus more on now at this point of your career?
I will say that I’m incredibly fortunate to have worked for as many years as I have in broadcast, and I’ve done some really amazing things, and had some phenomenal opportunities going to the Oscars and doing all those red carpets and celebrity interviews all over the world. They’re all valuable experiences that I’ve been able to build upon. But I will say that in the last sort of two years, especially, my focus has become more about Plus Life and doing these kinds of stories and these kinds of initiatives.
I am fascinated by people. I think to be a decent journalist or broadcaster, you have to have that innate curiosity. And I am deeply curious about people and people’s behavior and where they come from, and what kind of homes they grew up in, what their friends are like. It’s incredibly satisfying to me. Do I miss being on regular, scheduled programming and television? Sure. It was a fantastic platform, and there’s nothing quite like the thrill of live television for me, and that’s why I loved being in news and entertainment news.
I think as I sort of get older, and I’m no longer the young kid on the block, and I’m not so nimble with my thumbs and my forefingers when it comes to putting stuff up on social media and Tik Tok and all of that…If I’ve got an opportunity to really hone in on the stuff that I do with Plus Life, there’s nothing more rewarding than that, right?
A behind-the-scenes documentary of the U.S. debut of HIV Unwrapped will be available to stream on November 30th, just before World AIDS Day.
AIDS and HIV
Assemblymembers urge Governor Newsom to sign “lifeline” bill for HIV medication
AB 554 amends existing law that restricts access to PrEP medication

On September 10, Assembly Bill (AB) 554, or the PrEPARE Act (Protecting Rights, Expanding Prevention, and Advancing Reimbursement for Equity Act), was passed with a majority vote by the California State Assembly and Senate. Today, the bill’s leaders are urging Governor Newsom to sign it into law.
“With a powerful coalition behind AB 554, we were able to get this bill through the process swiftly, and are hopeful that the Governor will see the need for the LGBTQ+ community,” Los Angeles Assemblymember Mark Gonzalez, one of the bill’s co-authors, told the Blade.
AB 554 was first introduced in February, and was penned by Assemblymember Gonzalez and San Francisco Assemblymember Matt Haney. It was formed to amend existing laws around health insurance coverage that currently restrict access to certain antiretroviral drugs, or HIV preventative medicine like PrEP and PEP, and to ease difficulties community clinics face in administering these medications and receiving reimbursement. The bill will also require health care plans to cover FDA-approved HIV medication without enforcing prior authorization or step therapy.
For Gonzales and the bill’s supporters, AB 554 is about boosting access to various forms of effective HIV preventative medicine and providing a safeguard for people as the state of HIV research and treatment enters unsteady ground. This all comes in the midst of the debate around the House Committee on Appropriations’s 2026 Departments of Labor, Health and Human Services, and Education, and Related Agencies funding bill, which proposes $1.7 billion in cuts to domestic HIV prevention and research efforts, as well as programs that provide care for people living with HIV/AIDS — particularly, low-income people of color.
Assemblymember Gonzalez stated to the Blade that this is a “stark reminder that this epidemic still hits our communities of color the hardest,” and wants to address this by evolving and updating laws to reflect local needs. He is hopeful about the Governor’s impending response, and stated that the bill is “not just a policy; it is a lifeline.”
“It’s about giving people real choices, equipping small clinics with the tools they need to protect lives,” Gonzalez continued, “and ensuring that California continues to put public health over politics.”
AIDS and HIV
Local organization aims to support and assist Black LGBTQ+ community
REACH LA is stepping up their mission amid hostile administration

REACH LA, a Los Angeles-based nonprofit organization aimed at working with youth of color, is stepping up their prevention resources during Black History Month to support the LGBTQ+ community of color.
Though today is National Black HIV/AIDS Awareness Day, REACH LA works year-round to provide resources to their community members.
This month, the organization is amplifying its mission to support Black LGBTQ+ youth by offering free HIV testing and care throughout February, offering a $25 gift card as incentive to get tested. This and all of REACH LA’s efforts are geared toward assisting the marginalized Black and Latin American communities by reducing stigma, increasing education and assisting community members with resources.
The QTBIPOC community is especially vulnerable to political and personal attacks. As we head into the next four years under a hostile administration whose goal is to erase queer and trans people, there will be continued attacks on federal funding and on any other front possible.
“This year, it is especially vital, more than ever, to amplify and commemorate National Black HIV/AIDS Awareness Day. At REACH LA, we are currently engaging with individuals and partnerships while navigating through dire and uncertain times where HIV/AIDS awareness prevention efforts, access, and visibility have been under attack and restricted,” said Jeremiah Givens, chief marketing and communications officer at REACH LA.
It is important to spotlight the intersection between health equity, Black LGBTQ+ empowerment and community-based solutions during Black History Month and every other month throughout the year and especially during this particularly vulnerable time.
As one of eight CDC PACT Program Partners, REACH LA celebrates National Black HIV/AIDS Awareness Day with a Positive Living Campaign in collaboration with the CDC’s Let’s Stop HIV Together initiative. The campaign highlights the resilience of individuals living with HIV and works to raise awareness and foster community support.
To learn more about resources, visit their website or stop in for testing, support and other resources. The organization’s doors are open Monday through Friday, from 11 AM to 7 PM for free, on-site HIV testing and assistance with accessing PREP and PEP, linkage to care and free mental health therapy.

Over 300 people gathered last Monday to commemorate World AIDS Day in an event hosted by Bienestar Human Services. The non-profit focuses on identifying and addressing emerging health issues faced by Latinx and Lesbian, Gay, Bisexual, Trans Queer and more (LGBTQ+) populations.
The main objective of the annual event was to light a candle to honor those who passed away due to Human Immunodeficiency Virus (HIV) and celebrate those who, despite the condition, keep going.
HIV is the virus that causes Acquired Immune Deficiency Syndrome (AIDS). Once you have HIV, the virus stays in the body for life, and while there is no cure. There is, however, medicine that can help people stay healthy, states Planned Parenthood. HIV destroys the cells that protect the human body from infections. If a person doesn’t have enough of these CD4 or T cells, the body can’t fight off infections as normal. This damage can lead to AIDS, which is the most serious stage of HIV, and it leads to death over time.
In the U.S., there are about 1.3 million people newly infected with HIV in 2023 and 39.9 million living with HIV, according to the Joint United Nations Programme on HIV/AIDS.
Living with HIV for decades
Among the participants at the event at the Ukrainian Culture Center on Melrose in East Hollywood was Marcela, a transgender woman who was diagnosed HIV positive 33 years ago. She said when she learned about her diagnosis, she was so depressed she tried to commit suicide. She ended up at the hospital, and after getting better, she heard about an organization that was helping people with health issues such as hers.
“Since then, I learned to live with this condition because it is not an illness; it’s a condition,” she said.

Bienestar celebrated the World AIDS Day on December 2, 2024. (Courtesy of Bienestar)
Marcela, who didn’t provide her last name, said she not only found a new way of living with HIV but also found an extended family at Bienestar. They have guided her on how to receive the proper treatment and medicines, to be part of a support group and even to pay her rent when she needed it the most.
The 64-year-old woman doesn’t have any family members in the United States. She said five years ago, her husband passed away of pneumonia. She was living alone in Long Beach and got behind her payments during the pandemic. She said Bienestar helped her apply for a grant of over $20,000 that secured the payment for her rent, and now she is debt-free.
“I’m extremely grateful to them and all the help they provide,” she said.
Working for the community
This year, Bienestar is celebrating 35 years of serving the LGBTQ+ community. Among the many programs they offer is the Support Group for Transgender Women, which Marcela belongs to.
Mia Perez, the support group manager, said 15 to 20 members attend the group every Friday from 3 to 5 p.m. They talk about their feelings, share experiences, and plan and participate in social events.
Perez said one of the participants’ biggest concerns is accepting reality once they have been diagnosed. “However, with all these new treatments people that are HIV positive can have a normal relationship with someone who doesn’t have HIV. It’s all about getting informed,” said Perez.
Other concerns include what will happen to them once the new presidential administration takes office since they have plans to deport immigrants and many of the transgender women are immigrants.
“That’s why we are trying to get in contact with an immigration attorney or an organization so they can keep them informed of their rights,” said Perez.
Bienestar events, like World AIDS Day, help those affected create a stronger community, and they realize the recognition is not just for those who are HIV positive but also for their families and friends.
Marcela said when she is feeling down or bored at home, all she has to do is go to Bienestar and the people there always give her a warm welcome.
“They give me coffee, they offer me lunch. Being there is like being at home,” she said.
-

 Hungary5 days ago
Hungary5 days agoHungarian authorities lift Budapest Pride ban
-

 Pride Special4 days ago
Pride Special4 days agoControversy looms as Kathy Hilton is named West Hollywood Pride Grand Marshal
-

 Celebrity News3 days ago
Celebrity News3 days agoOutright International honors Cyndi Lauper at annual NYC gala
-

 Theater4 days ago
Theater4 days agoIn ‘A Man Called Mommy,’ Noadiah Eckman delivers queer joy across the aisle
-

 Congress4 days ago
Congress4 days ago10 HIV/AIDS activists arrested on Capitol Hill
-

 Politics3 days ago
Politics3 days agoLos Angeles Primary Election Day results are in
-

 Politics5 days ago
Politics5 days agoFrom the desk of Equality California: The legal issues hitting California and beyond this Pride Month
-

 Federal Government4 days ago
Federal Government4 days agoAdvocates push back on proposed FCC warning labels
-

 Film5 days ago
Film5 days ago‘AIDS DIVA’ screening honors Connie Norman’s legacy
-

 LGBTQ Non-Profit Organizations5 days ago
LGBTQ Non-Profit Organizations5 days agoPride is thriving at the LA LGBT Center











